Comparative experimental subcutaneous glanders and melioidosis in the common marmoset (Callithrix jacchus)
- PMID: 25477002
- PMCID: PMC4285464
- DOI: 10.1111/iep.12105
Comparative experimental subcutaneous glanders and melioidosis in the common marmoset (Callithrix jacchus)
Abstract
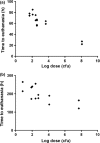
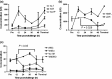
Glanders and melioidosis are caused by two distinct Burkholderia species and have generally been considered to have similar disease progression. While both of these pathogens are HHS/CDC Tier 1 agents, natural infection with both these pathogens is primarily through skin inoculation. The common marmoset (Callithrix jacchus) was used to compare disease following experimental subcutaneous challenge. Acute, lethal disease was observed in marmosets following challenge with between 26 and 1.2 × 10(8) cfu Burkholderia pseudomallei within 22-85 h. The reproducibility and progression of the disease were assessed following a challenge of 1 × 10(2) cfu of B. pseudomallei. Melioidosis was characterised by high levels of bacteraemia, focal microgranuloma progressing to non-necrotic multifocal solid lesions in the livers and spleens and multi-organ failure. Lethal disease was observed in 93% of animals challenged with Burkholderia mallei, occurring between 5 and 10.6 days. Following challenge with 1 × 10(2) cfu of B. mallei, glanders was characterised with lymphatic spread of the bacteria and non-necrotic, multifocal solid lesions progressing to a multifocal lesion with severe necrosis and pneumonia. The experimental results confirmed that the disease pathology and presentation is strikingly different between the two pathogens. The marmoset provides a model of the human syndrome for both diseases facilitating the development of medical countermeasures.
Keywords: Burkholderia; animal model; histology.
© 2014 Crown copyright. International Journal of Experimental Pathology © 2014 Company of the International Journal of Experimental Pathology (CIJEP).
Figures





Similar articles
-
Characterization of lesion formation in marmosets following inhalational challenge with different strains of Burkholderia pseudomallei.Int J Exp Pathol. 2015 Dec;96(6):414-26. doi: 10.1111/iep.12161. Epub 2016 Jan 19. Int J Exp Pathol. 2015. PMID: 26852689 Free PMC article.
-
Molecular insights into Burkholderia pseudomallei and Burkholderia mallei pathogenesis.Annu Rev Microbiol. 2010;64:495-517. doi: 10.1146/annurev.micro.112408.134030. Annu Rev Microbiol. 2010. PMID: 20528691 Review.
-
Use of a safe, reproducible, and rapid aerosol delivery method to study infection by Burkholderia pseudomallei and Burkholderia mallei in mice.PLoS One. 2013 Oct 2;8(10):e76804. doi: 10.1371/journal.pone.0076804. eCollection 2013. PLoS One. 2013. PMID: 24098563 Free PMC article.
-
Use of the common marmoset to study Burkholderia mallei infection.PLoS One. 2015 Apr 10;10(4):e0124181. doi: 10.1371/journal.pone.0124181. eCollection 2015. PLoS One. 2015. PMID: 25860021 Free PMC article.
-
Burkholderia mallei and Burkholderia pseudomallei: the causative micro-organisms of glanders and melioidosis.Recent Pat Antiinfect Drug Discov. 2007 Nov;2(3):233-41. doi: 10.2174/157489107782497335. Recent Pat Antiinfect Drug Discov. 2007. PMID: 18221181 Review.
Cited by
-
The lymphatic system as a potential mechanism of spread of melioidosis following ingestion of Burkholderia pseudomallei.PLoS Negl Trop Dis. 2021 Feb 22;15(2):e0009016. doi: 10.1371/journal.pntd.0009016. eCollection 2021 Feb. PLoS Negl Trop Dis. 2021. PMID: 33617546 Free PMC article.
-
Dose-dependant acute or subacute disease caused by Burkholderia pseudomallei strain NCTC 13392 in a BALB/c aerosol model of infection.J Appl Microbiol. 2019 Oct;127(4):1224-1235. doi: 10.1111/jam.14396. Epub 2019 Aug 9. J Appl Microbiol. 2019. PMID: 31330088 Free PMC article.
-
Novel multi-component vaccine approaches for Burkholderia pseudomallei.Clin Exp Immunol. 2019 May;196(2):178-188. doi: 10.1111/cei.13286. Epub 2019 Apr 8. Clin Exp Immunol. 2019. PMID: 30963550 Free PMC article. Review.
-
Consensus on the development of vaccines against naturally acquired melioidosis.Emerg Infect Dis. 2015 Jun;21(6):e141480. doi: 10.3201/eid2106.141480. Emerg Infect Dis. 2015. PMID: 25992835 Free PMC article.
-
Characterization of lesion formation in marmosets following inhalational challenge with different strains of Burkholderia pseudomallei.Int J Exp Pathol. 2015 Dec;96(6):414-26. doi: 10.1111/iep.12161. Epub 2016 Jan 19. Int J Exp Pathol. 2015. PMID: 26852689 Free PMC article.
References
-
- Currie BJ, Fisher DA, Howard DM, et al. The epidemiology of melioidosis in Australia and Papua New Guinea. Acta Trop. 2000;74:121–127. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

