Mitochondrial dynamics and mitochondrial quality control
- PMID: 25479550
- PMCID: PMC4309858
- DOI: 10.1016/j.redox.2014.11.006
Mitochondrial dynamics and mitochondrial quality control
Abstract
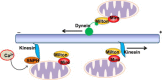
Mitochondria are cellular energy powerhouses that play important roles in maintaining cell survival, cell death and cellular metabolic homeostasis. Timely removal of damaged mitochondria via autophagy (mitophagy) is thus critical for cellular homeostasis and function. Mitochondria are reticular organelles that have high plasticity for their dynamic structures and constantly undergo fission and fusion as well as movement through the cytoskeleton. In this review, we discuss the most recent progress on the molecular mechanisms and roles of mitochondrial fission/fusion and mitochondrial motility in mitophagy. We also discuss multiple pathways leading to the quality control of mitochondria in addition to the traditional mitophagy pathway under different conditions.
Keywords: Autophagy; Mitochondrial spheroids; Mitophagy; Parkin.
Copyright © 2014 The Authors. Published by Elsevier B.V. All rights reserved.
Figures





References
Publication types
MeSH terms
Grants and funding
- R01 DK102142/DK/NIDDK NIH HHS/United States
- R01DK102142/DK/NIDDK NIH HHS/United States
- P20 GM103549/GM/NIGMS NIH HHS/United States
- P20 GM103418/GM/NIGMS NIH HHS/United States
- 5P20RR021940/RR/NCRR NIH HHS/United States
- P30 AG035982/AG/NIA NIH HHS/United States
- T32 ES007079/ES/NIEHS NIH HHS/United States
- R01 AA020518/AA/NIAAA NIH HHS/United States
- R01AA020518/AA/NIAAA NIH HHS/United States
- 8P20GM103549/GM/NIGMS NIH HHS/United States
- P20GM103418/GM/NIGMS NIH HHS/United States
- R37 AA020518/AA/NIAAA NIH HHS/United States
- P20 RR021940/RR/NCRR NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources

