Intravenous and gastric cerium dioxide nanoparticle exposure disrupts microvascular smooth muscle signaling
- PMID: 25481005
- PMCID: PMC4349137
- DOI: 10.1093/toxsci/kfu256
Intravenous and gastric cerium dioxide nanoparticle exposure disrupts microvascular smooth muscle signaling
Abstract
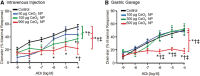
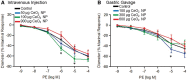
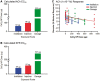
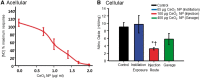
Cerium dioxide nanoparticles (CeO2 NP) hold great therapeutic potential, but the in vivo effects of non-pulmonary exposure routes are unclear. The first aim was to determine whether microvascular function is impaired after intravenous and gastric CeO2 NP exposure. The second aim was to investigate the mechanism(s) of action underlying microvascular dysfunction following CeO2 NP exposure. Rats were exposed to CeO2 NP (primary diameter: 4 ± 1 nm, surface area: 81.36 m(2)/g) by intratracheal instillation, intravenous injection, or gastric gavage. Mesenteric arterioles were harvested 24 h post-exposure and vascular function was assessed using an isolated arteriole preparation. Endothelium-dependent and independent function and vascular smooth muscle (VSM) signaling (soluble guanylyl cyclase [sGC] and cyclic guanosine monophosphate [cGMP]) were assessed. Reactive oxygen species (ROS) generation and nitric oxide (NO) production were analyzed. Compared with controls, endothelium-dependent and independent dilation were impaired following intravenous injection (by 61% and 45%) and gastric gavage (by 63% and 49%). However, intravenous injection resulted in greater microvascular impairment (16% and 35%) compared with gastric gavage at an identical dose (100 µg). Furthermore, sGC activation and cGMP responsiveness were impaired following pulmonary, intravenous, and gastric CeO2 NP treatment. Finally, nanoparticle exposure resulted in route-dependent, increased ROS generation and decreased NO production. These results indicate that CeO2 NP exposure route differentially impairs microvascular function, which may be mechanistically linked to decreased NO production and subsequent VSM signaling. Fully understanding the mechanisms behind CeO2 NP in vivo effects is a critical step in the continued therapeutic development of this nanoparticle.
Keywords: cerium dioxide nanoparticles; microvascular function; nitric oxide; mesentery.
© The Author 2014. Published by Oxford University Press on behalf of the Society of Toxicology. All rights reserved. For Permissions, please e-mail: journals.permissions@oup.com.
Figures






Similar articles
-
Pulmonary cerium dioxide nanoparticle exposure differentially impairs coronary and mesenteric arteriolar reactivity.Cardiovasc Toxicol. 2013 Dec;13(4):323-37. doi: 10.1007/s12012-013-9213-3. Cardiovasc Toxicol. 2013. PMID: 23645470 Free PMC article.
-
Regulation of the expression of soluble guanylyl cyclase by reactive oxygen species.Br J Pharmacol. 2007 Apr;150(8):1084-91. doi: 10.1038/sj.bjp.0707179. Epub 2007 Mar 5. Br J Pharmacol. 2007. PMID: 17339839 Free PMC article.
-
Cerium Dioxide Nanoparticle Exposure Improves Microvascular Dysfunction and Reduces Oxidative Stress in Spontaneously Hypertensive Rats.Front Physiol. 2015 Nov 17;6:339. doi: 10.3389/fphys.2015.00339. eCollection 2015. Front Physiol. 2015. PMID: 26635625 Free PMC article.
-
Nitric oxide-independent stimulation of soluble guanylate cyclase with BAY 41-2272 in cardiovascular disease.Cardiovasc Drug Rev. 2007 Spring;25(1):30-45. doi: 10.1111/j.1527-3466.2007.00003.x. Cardiovasc Drug Rev. 2007. PMID: 17445086 Review.
-
Structure and regulation of soluble guanylate cyclase.Annu Rev Biochem. 2012;81:533-59. doi: 10.1146/annurev-biochem-050410-100030. Epub 2012 Feb 9. Annu Rev Biochem. 2012. PMID: 22404633 Review.
Cited by
-
Vasomotor function in rat arteries after ex vivo and intragastric exposure to food-grade titanium dioxide and vegetable carbon particles.Part Fibre Toxicol. 2018 Feb 26;15(1):12. doi: 10.1186/s12989-018-0248-2. Part Fibre Toxicol. 2018. PMID: 29482579 Free PMC article.
-
Nanoparticle Effects on Stress Response Pathways and Nanoparticle-Protein Interactions.Int J Mol Sci. 2022 Jul 19;23(14):7962. doi: 10.3390/ijms23147962. Int J Mol Sci. 2022. PMID: 35887304 Free PMC article. Review.
-
Engineered nanomaterials and oxidative stress: current understanding and future challenges.Curr Opin Toxicol. 2019 Feb;13:74-80. doi: 10.1016/j.cotox.2018.09.001. Epub 2018 Sep 7. Curr Opin Toxicol. 2019. PMID: 31263794 Free PMC article.
-
Metal Nanomaterial Toxicity Variations Within the Vascular System.Curr Environ Health Rep. 2016 Dec;3(4):379-391. doi: 10.1007/s40572-016-0112-1. Curr Environ Health Rep. 2016. PMID: 27686080 Free PMC article. Review.
-
Lung versus gut exposure to air pollution particles differentially affect metabolic health in mice.Part Fibre Toxicol. 2023 Mar 9;20(1):7. doi: 10.1186/s12989-023-00518-w. Part Fibre Toxicol. 2023. PMID: 36895000 Free PMC article.
References
-
- Boger R. H., Bode-Boger S. M. (2000). Asymmetric dimethylarginine, derangements of the endothelial nitric oxide synthase pathway, and cardiovascular diseases. Semin. Thromb. Hemost. 26, 539–545. - PubMed
-
- Brannon-Peppas L., Blanchette J. O. (2004). Nanoparticle and targeted systems for cancer therapy. Adv. Drug Deliv. Rev. 56, 1649–1659. - PubMed
-
- Buettner G. R. (1987). Spin trapping: ESR parameters of spin adducts. Free Radic. Biol. Med. 3, 259–303. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

