Conditionally activating optical contrast agent with enhanced sensitivity via gold nanoparticle plasmon energy transfer: feasibility study
- PMID: 25481683
- PMCID: PMC4267155
- DOI: 10.1186/s12951-014-0056-2
Conditionally activating optical contrast agent with enhanced sensitivity via gold nanoparticle plasmon energy transfer: feasibility study
Abstract
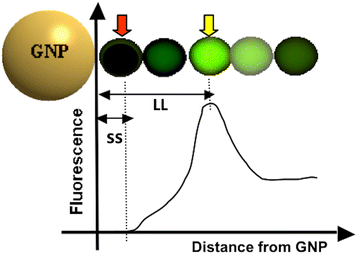
Background: Molecular sensing/imaging utilizing fluorophores has been one of the most frequently used techniques in biomedical research. As for any molecular imaging techniques, fluorescence mediated sensing always seeks for greater specificity and sensitivity. Since fluorophores emit fluorescence while their electron energy state changes, manipulating the local electromagnetic field around the fluorophores may be a way to enhance the specificity and sensitivity. Gold nanoparticles (GNPs) are known to form a very strong electromagnetic field on their surface [i.e., surface plasmon field (SPF)], upon receiving photonic energy. The level of fluorescence change by GNP-SPF may range from complete quenching to extensive enhancement, depending upon the SPF strength, excitation and emission wavelengths, and quantum yield of the fluorophore.
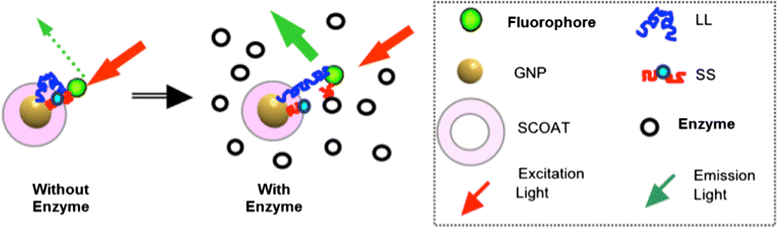
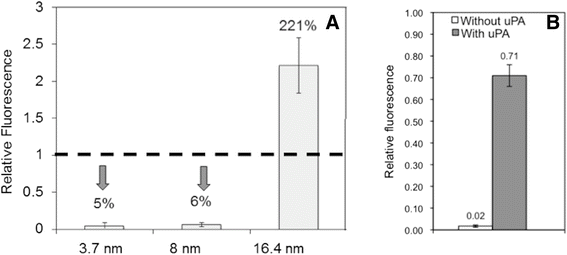
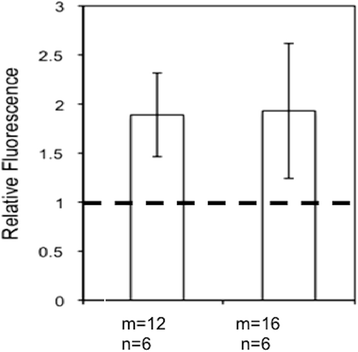
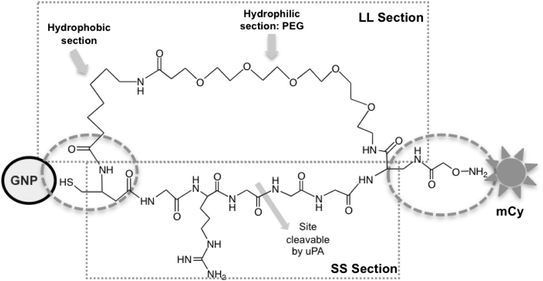
Method: Here, we report a novel design that utilizes BOTH fluorescence quenching and enhancement abilities of the GNP in one single nano-entity, providing high specificity and sensitivity. The construct utilizes a specially designed molecular dual-spacer that places the fluorphore at the location with an appropriate GNP-SFP strength before and after exposed to the biomarker. A model system to test the concept was an optical signal mediator activated by urokinase-type plasminogen activator (uPA; breast cancer secreting enzyme).
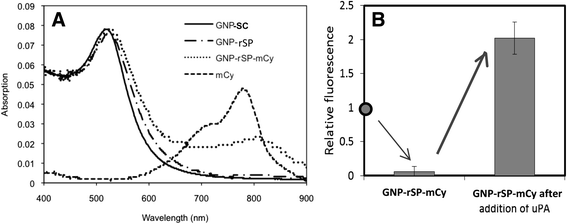
Results: The resulting contrast agent shows less than 10% of the natural fluorescence but, in the presence of uPA, its fluorescence emission is triggered and emits its fluorescence approximately twice of the natural form.
Conclusion: This study demonstrated that our novel design of an optical contrast agent can be conditionally activated with enhanced sensitivity, using both quenching and enhancement phenomena of fluorophores in the electromagnetic field of the appropriate strengths (in this case, locally generated by the GNP-SPF). This entity is similar to molecular beacon in terms of specificity but with greater sensitivity. In addition, it is not restricted to only DNA or RNA sensing but for any designs that cause the change in the distance between the fluorophore and GNP, upon the time of encountering biomarker of interest.
Figures



Similar articles
-
Gold nanoparticle-fluorophore complex for conditionally fluorescing signal mediator.Anal Chim Acta. 2011 Jun 10;695(1-2):96-104. doi: 10.1016/j.aca.2011.03.058. Epub 2011 Apr 7. Anal Chim Acta. 2011. PMID: 21601036
-
Fluorophore-gold nanoparticle complex for sensitive optical biosensing and imaging.Nanotechnology. 2012 Mar 9;23(9):095501. doi: 10.1088/0957-4484/23/9/095501. Epub 2012 Feb 10. Nanotechnology. 2012. PMID: 22327387
-
Fluorescence manipulation by gold nanoparticles: from complete quenching to extensive enhancement.J Nanobiotechnology. 2011 May 10;9:16. doi: 10.1186/1477-3155-9-16. J Nanobiotechnology. 2011. PMID: 21569249 Free PMC article.
-
Smart dual-mode fluorescent gold nanoparticle agents.Wiley Interdiscip Rev Nanomed Nanobiotechnol. 2014 Jul-Aug;6(4):398-409. doi: 10.1002/wnan.1267. Epub 2014 Apr 8. Wiley Interdiscip Rev Nanomed Nanobiotechnol. 2014. PMID: 24715524 Review.
-
Exploiting the light-metal interaction for biomolecular sensing and imaging.Q Rev Biophys. 2012 May;45(2):209-55. doi: 10.1017/S0033583512000042. Q Rev Biophys. 2012. PMID: 22559015 Review.
Cited by
-
Recent advances in light-responsive on-demand drug-delivery systems.Ther Deliv. 2017 Feb;8(2):89-107. doi: 10.4155/tde-2016-0060. Ther Deliv. 2017. PMID: 28088880 Free PMC article. Review.
References
-
- Zayats AV, Smolyaninov II, Maradudin AA. Nano-optics of surface plasmon polaritons. Physics Rep-Rev Sect Phys Lett. 2005;408:131–314.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

