The basic N-terminal domain of TRF2 limits recombination endonuclease action at human telomeres
- PMID: 25483196
- PMCID: PMC4128890
- DOI: 10.4161/cc.29422
The basic N-terminal domain of TRF2 limits recombination endonuclease action at human telomeres
Abstract
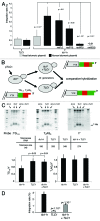
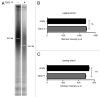
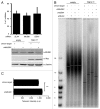
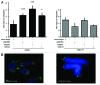
The stability of mammalian telomeres depends upon TRF2, which prevents inappropriate repair and checkpoint activation. By using a plasmid integration assay in yeasts carrying humanized telomeres, we demonstrated that TRF2 possesses the intrinsic property to both stimulate initial homologous recombination events and to prevent their resolution via its basic N-terminal domain. In human cells, we further showed that this TRF2 domain prevents telomere shortening mediated by the resolvase-associated protein SLX4 as well as GEN1 and MUS81, 2 different types of endonucleases with resolvase activities. We propose that various types of resolvase activities are kept in check by the basic N-terminal domain of TRF2 in order to favor an accurate repair of the stalled forks that occur during telomere replication.
Keywords: TRF2; recombination; shelterin; telomere.
Figures

References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous
