A novel crosstalk between BRCA1 and poly (ADP-ribose) polymerase 1 in breast cancer
- PMID: 25485588
- PMCID: PMC4613991
- DOI: 10.4161/15384101.2014.956507
A novel crosstalk between BRCA1 and poly (ADP-ribose) polymerase 1 in breast cancer
Abstract
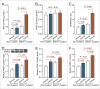
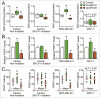
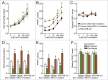
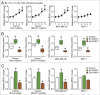
BRCA mutations are the main known hereditary factor for breast cancer. Notably, poly (ADP-ribose) polymerase 1 (PARP1) expression status plays a critical role in breast cancer progression and the clinical development of PARP1 inhibitors to treat BRCA-mutated breast cancer has advanced rapidly. However, dynamic crosstalk between BRCA1 and PARP1 remains largely unknown. Here, we showed that: (i) BRCA1 inactivation events (mutation, promoter methylation, or knockdown) were accompanied by increased PARP1 and nicotinamide adenine dinucleotide (NAD) levels, and a subsequent increase in NAD-dependent PARP1 activity in MDA-MB-231 and primary breast cancer cells; (ii) the overexpression of BRCA1 resulted in decreased PARP1 and NAD levels, and a subsequent impairment in NAD-dependent PARP1 activity in MDA-MB-231 and primary breast cancer cells; and (iii) intracellular NAD levels were largely responsible for regulating PARP1 activity in breast cancer cells, and NAD levels were positively correlated with PARP1 activity in human breast cancer specimens (R = 0.647, P < 0.001). Interestingly, the high efficiency of PARP1 triggered by BRCA1 inactivation may further inhibit BRCA1 transcription by NAD depletion. These results highlight a novel interaction between BRCA1 and PARP1, which may be beneficial for the dynamic balance between BRCA1 and PARP1-related biologic processes, especially for maintaining stable DNA repair ability. All of this may improve our understanding of the basic molecular mechanism underlying BRCA1- and PARP1-related breast cancer progression.
Keywords: BRCA1; CtBP, C-terminal binding proteins; DMEM, Dulbecco's Modified Eagles Medium; DNA repair; ER, endoplasmic reticulum; ETS1, protein C-ets-1; NAD; NAD, nicotinamide adenine dinucleotide; Nampt, nicotinamide phosphoribosyltransferase; PARP1; PARP1, poly (ADP-ribose) polymerase 1; PCR, polymerase chain reaction; SD, standard deviations; TNBC, triple-negative breast cancer; breast cancer; shRNAs, short hairpin RNAs.
Figures

Similar articles
-
BRCA1 as a nicotinamide adenine dinucleotide (NAD)-dependent metabolic switch in ovarian cancer.Cell Cycle. 2014;13(16):2564-71. doi: 10.4161/15384101.2015.942208. Cell Cycle. 2014. PMID: 25486197 Free PMC article.
-
Promoter hypomethylation, especially around the E26 transformation-specific motif, and increased expression of poly (ADP-ribose) polymerase 1 in BRCA-mutated serous ovarian cancer.BMC Cancer. 2013 Feb 26;13:90. doi: 10.1186/1471-2407-13-90. BMC Cancer. 2013. PMID: 23442605 Free PMC article.
-
Biological and clinical significance of PARP1 protein expression in breast cancer.Breast Cancer Res Treat. 2015 Jan;149(2):353-62. doi: 10.1007/s10549-014-3230-1. Epub 2014 Dec 21. Breast Cancer Res Treat. 2015. PMID: 25528020 Free PMC article.
-
BRCA Gene Mutations and Poly(ADP-Ribose) Polymerase Inhibitors in Triple-Negative Breast Cancer.Adv Exp Med Biol. 2017;1026:271-286. doi: 10.1007/978-981-10-6020-5_13. Adv Exp Med Biol. 2017. PMID: 29282689 Review.
-
Poly(ADP-ribose) Polymerase (PARP) and PARP Inhibitors: Mechanisms of Action and Role in Cardiovascular Disorders.Cardiovasc Toxicol. 2018 Dec;18(6):493-506. doi: 10.1007/s12012-018-9462-2. Cardiovasc Toxicol. 2018. PMID: 29968072 Review.
Cited by
-
Characterization of Parthanatos in Breast Cancer: Implications for Prognosis and PARP Inhibitor Resistance.Bioengineering (Basel). 2025 May 29;12(6):586. doi: 10.3390/bioengineering12060586. Bioengineering (Basel). 2025. PMID: 40564403 Free PMC article.
-
Nicotinamide adenine dinucleotide (NAD) may affect DNA methyltransferase 1 through regulation of BRCA1 in ovarian cancer.Am J Cancer Res. 2015 Feb 15;5(3):1199-206. eCollection 2015. Am J Cancer Res. 2015. PMID: 26045998 Free PMC article.
-
Nicotinamide is an inhibitor of SIRT1 in vitro, but can be a stimulator in cells.Cell Mol Life Sci. 2017 Sep;74(18):3347-3362. doi: 10.1007/s00018-017-2527-8. Epub 2017 Apr 17. Cell Mol Life Sci. 2017. PMID: 28417163 Free PMC article. Review.
-
Mutated Fanconi anemia pathway in non-Fanconi anemia cancers.Oncotarget. 2015 Aug 21;6(24):20396-403. doi: 10.18632/oncotarget.4056. Oncotarget. 2015. PMID: 26015400 Free PMC article.
-
NetCellMatch: Multiscale Network-Based Matching of Cancer Cell Lines to Patients Using Graphical Wavelets.Chem Biodivers. 2022 Dec;19(12):e202200746. doi: 10.1002/cbdv.202200746. Epub 2022 Nov 28. Chem Biodivers. 2022. PMID: 36279370 Free PMC article.
References
-
- DeSantis C, Ma J, Bryan L, Jemal A. Breast cancer statistics, 2013. CA Cancer J Clin 2014; 64:52-62; PMID:24114568; http://dx.doi.org/10.3322/caac.21203 - DOI - PubMed
-
- Pruthi S, Gostout BS, Lindor NM. Identification and management of women Wwth BRCA mutations or hereditary predisposition for breast and ovarian cancer. Mayo Clin Proc 2010; 85:1111-20; PMID:21123638; http://dx.doi.org/10.4065/mcp.2010.0414 - DOI - PMC - PubMed
-
- Bryant HE, Schultz N, Thomas HD, Parker KM, Flower D, Lopez E, Kyle S, Meuth M, Curtin NJ, Helleday T. Specific killing of BRCA2-deficient tumours with inhibitors of poly(ADP-ribose) polymerase. Nature 2005; 434:913-7; PMID:15829966; http://dx.doi.org/10.1038/nature03443 - DOI - PubMed
-
- Farmer H, McCabe N, Lord CJ, Tutt AN, Johnson DA, Richardson TB, Santarosa M, Dillon KJ, Hickson I, Knights C, et al. . Targeting the DNA repair defect in BRCA mutant cells as a therapeutic strategy. Nature 2005; 434:917-21; PMID:15829967; http://dx.doi.org/10.1038/nature03445 - DOI - PubMed
-
- Dacheux E, Vincent A, Nazaret N, Combet C, Wierinckx A, Mazoyer S, Diaz JJ, Lachuer J, Venezia ND. BRCA1-dependent translational regulation in breast cancer cells. PLoS One 2013; 8:e67313; PMID: 23805307; http://dx.doi.org/10.1371/journal.pone.0067313 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous
