ZNF300 knockdown inhibits forced megakaryocytic differentiation by phorbol and erythrocytic differentiation by arabinofuranosyl cytidine in K562 cells
- PMID: 25485965
- PMCID: PMC4259388
- DOI: 10.1371/journal.pone.0114768
ZNF300 knockdown inhibits forced megakaryocytic differentiation by phorbol and erythrocytic differentiation by arabinofuranosyl cytidine in K562 cells
Abstract
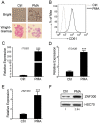
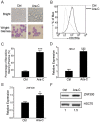
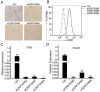
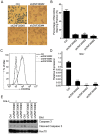
Previously, we reported that ZNF300 might play a role in leukemogenesis. In this study, we further investigated the function of ZNF300 in K562 cells undergoing differentiation. We found that ZNF300 upregulation in K562 cells coincided with megakaryocytic differentiation induced by phorbol-12-myristate-13-acetate (PMA) or erythrocytic differentiation induced by cytosine arabinoside (Ara-C), respectively. To further test whether ZNF300 upregulation promoted differentiation, we knocked down ZNF300 and found that ZNF300 knockdown effectively abolished PMA-induced megakaryocytic differentiation, evidenced by decreased CD61 expression. Furthermore, Ara-C-induced erythrocytic differentiation was also suppressed in ZNF300 knockdown cells with decreased γ-globin expression and CD235a expression. These observations suggest that ZNF300 may be a critical factor controlling distinct aspects of K562 cells. Indeed, ZNF300 knockdown led to increased cell proliferation. Consistently, ZNF300 knockdown cells exhibited an increased percentage of cells at S phase accompanied by decreased percentage of cells at G0/G1 and G2/M phase. Increased cell proliferation was further supported by the increased expression of cell proliferation marker PCNA and the decreased expression of cell cycle regulator p15 and p27. In addition, MAPK/ERK signaling was significantly suppressed by ZNF300 knockdown. These findings suggest a potential mechanism by which ZNF300 knockdown may impair megakaryocytic and erythrocytic differentiation.
Conflict of interest statement
Figures
Similar articles
-
[ZNF300 Promotes Megakaryocytic Differentiation].Zhongguo Shi Yan Xue Ye Xue Za Zhi. 2017 Oct;25(5):1537-1543. doi: 10.7534/j.issn.1009-2137.2017.05.046. Zhongguo Shi Yan Xue Ye Xue Za Zhi. 2017. PMID: 29070140 Chinese.
-
Protein-arginine methyltransferase 1 suppresses megakaryocytic differentiation via modulation of the p38 MAPK pathway in K562 cells.J Biol Chem. 2010 Jul 2;285(27):20595-606. doi: 10.1074/jbc.M109.092411. Epub 2010 May 4. J Biol Chem. 2010. PMID: 20442406 Free PMC article.
-
Nobiletin Promotes Megakaryocytic Differentiation through the MAPK/ERK-Dependent EGR1 Expression and Exerts Anti-Leukemic Effects in Human Chronic Myeloid Leukemia (CML) K562 Cells.Cells. 2020 Apr 3;9(4):877. doi: 10.3390/cells9040877. Cells. 2020. PMID: 32260160 Free PMC article.
-
Membrane type sialidase inhibits the megakaryocytic differentiation of human leukemia K562 cells.Biochim Biophys Acta. 2008 May;1780(5):757-63. doi: 10.1016/j.bbagen.2008.01.019. Epub 2008 Feb 20. Biochim Biophys Acta. 2008. PMID: 18339327
-
Differential expression changes in K562 cells during the hemin-induced erythroid differentiation and the phorbol myristate acetate (PMA)-induced megakaryocytic differentiation.Mol Cell Biochem. 2006 Nov;292(1-2):155-67. doi: 10.1007/s11010-006-9229-0. Epub 2006 Jun 20. Mol Cell Biochem. 2006. PMID: 16786195
Cited by
-
KRAB-ZFP Transcriptional Regulators Acting as Oncogenes and Tumor Suppressors: An Overview.Int J Mol Sci. 2021 Feb 23;22(4):2212. doi: 10.3390/ijms22042212. Int J Mol Sci. 2021. PMID: 33672287 Free PMC article. Review.
-
Novel function of FAXDC2 in megakaryopoiesis.Blood Cancer J. 2016 Sep 30;6(9):e478. doi: 10.1038/bcj.2016.87. Blood Cancer J. 2016. PMID: 27689744 Free PMC article.
-
Genome-wide methylation sequencing identifies progression-related epigenetic drivers in myelodysplastic syndromes.Cell Death Dis. 2020 Nov 20;11(11):997. doi: 10.1038/s41419-020-03213-2. Cell Death Dis. 2020. PMID: 33219204 Free PMC article.
-
Genome-wide placental DNA methylation analysis of severely growth-discordant monochorionic twins reveals novel epigenetic targets for intrauterine growth restriction.Clin Epigenetics. 2016 Jun 21;8:70. doi: 10.1186/s13148-016-0238-x. eCollection 2016. Clin Epigenetics. 2016. PMID: 27330572 Free PMC article.
-
Detecting serum and urine metabolic profile changes of CCl4-liver fibrosis in rats at 12 weeks based on gas chromatography-mass spectrometry.Exp Ther Med. 2017 Aug;14(2):1496-1504. doi: 10.3892/etm.2017.4668. Epub 2017 Jun 26. Exp Ther Med. 2017. PMID: 28810615 Free PMC article.
References
-
- Agata Y, Matsuda E, Shimizu A (1999) Two novel Kruppel-associated box-containing zinc-finger proteins, KRAZ1 and KRAZ2, repress transcription through functional interaction with the corepressor KAP-1 (TIF1beta/KRIP-1). J Biol Chem 274:16412–16422. - PubMed
-
- Wanda PE, Walker MM (1989) Hemoglobin induction by Ara-C in human erythroleukemic cells (K562) is cell-cycle dependent. Leuk Res 13:683–688. - PubMed
-
- Ryan RF, Schultz DC, Ayyanathan K, Singh PB, Friedman JR, et al. (1999) KAP-1 corepressor protein interacts and colocalizes with heterochromatic and euchromatic HP1 proteins: a potential role for Kruppel-associated box-zinc finger proteins in heterochromatin-mediated gene silencing. Mol Cell Biol 19:4366–4378. - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

