Mycobacterium abscessus phospholipase C expression is induced during coculture within amoebae and enhances M. abscessus virulence in mice
- PMID: 25486995
- PMCID: PMC4294255
- DOI: 10.1128/IAI.02032-14
Mycobacterium abscessus phospholipase C expression is induced during coculture within amoebae and enhances M. abscessus virulence in mice
Abstract
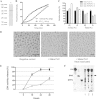
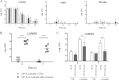
Mycobacterium abscessus is a pathogenic, rapidly growing mycobacterium involved in pulmonary and cutaneo-mucous infections worldwide, to which cystic fibrosis patients are exquisitely susceptible. The analysis of the genome sequence of M. abscessus showed that this bacterium is endowed with the metabolic pathways typically found in environmental microorganisms that come into contact with soil, plants, and aquatic environments, where free-living amoebae are frequently present. M. abscessus also contains several genes that are characteristically found only in pathogenic bacteria. One of them is MAB_0555, encoding a putative phospholipase C (PLC) that is absent from most other rapidly growing mycobacteria, including Mycobacterium chelonae and Mycobacterium smegmatis. Here, we report that purified recombinant M. abscessus PLC is highly cytotoxic to mouse macrophages, presumably due to hydrolysis of membrane phospholipids. We further showed by constructing and using an M. abscessus PLC knockout mutant that loss of PLC activity is deleterious to M. abscessus intracellular survival in amoebae. The importance of PLC is further supported by the fact that M. abscessus PLC was found to be expressed only in amoebae. Aerosol challenge of mice with M. abscessus strains that were precultured in amoebae enhanced M. abscessus lung infectivity relative to M. abscessus grown in broth culture. Our study underlines the importance of PLC for the virulence of M. abscessus. Despite the difficulties of isolating M. abscessus from environmental sources, our findings suggest that M. abscessus has evolved in close contact with environmental protozoa, which supports the argument that amoebae may contribute to the virulence of opportunistic mycobacteria.
Copyright © 2015, American Society for Microbiology. All Rights Reserved.
Figures


Similar articles
-
Bacterial phospholipases C as vaccine candidate antigens against cystic fibrosis respiratory pathogens: the Mycobacterium abscessus model.Vaccine. 2015 Apr 27;33(18):2118-24. doi: 10.1016/j.vaccine.2015.03.030. Epub 2015 Mar 21. Vaccine. 2015. PMID: 25804706
-
Mycobacterium abscessus virulence traits unraveled by transcriptomic profiling in amoeba and macrophages.PLoS Pathog. 2019 Nov 8;15(11):e1008069. doi: 10.1371/journal.ppat.1008069. eCollection 2019 Nov. PLoS Pathog. 2019. PMID: 31703112 Free PMC article.
-
MgtC as a Host-Induced Factor and Vaccine Candidate against Mycobacterium abscessus Infection.Infect Immun. 2016 Sep 19;84(10):2895-903. doi: 10.1128/IAI.00359-16. Print 2016 Oct. Infect Immun. 2016. PMID: 27481243 Free PMC article.
-
Mycobacterium abscessus, a complex of three fast-growing subspecies sharing virulence traits with slow-growing mycobacteria.Clin Microbiol Infect. 2024 Jun;30(6):726-731. doi: 10.1016/j.cmi.2023.08.036. Epub 2023 Oct 4. Clin Microbiol Infect. 2024. PMID: 37797823 Review.
-
Mechanisms of Virulence of Mycobacterium abscessus and Interaction with the Host Immune System.Biochemistry (Mosc). 2025 Jan;90(Suppl 1):S214-S232. doi: 10.1134/S0006297924603496. Biochemistry (Mosc). 2025. PMID: 40164160 Review.
Cited by
-
Host-Pathogen Interactions Operative during Mycobacteroides abscessus Infection.Immune Netw. 2021 Dec 23;21(6):e40. doi: 10.4110/in.2021.21.e40. eCollection 2021 Dec. Immune Netw. 2021. PMID: 35036027 Free PMC article. Review.
-
Vaccine strategies against cystic fibrosis pathogens.Hum Vaccin Immunother. 2016 Mar 3;12(3):751-6. doi: 10.1080/21645515.2015.1102810. Hum Vaccin Immunother. 2016. PMID: 26618824 Free PMC article.
-
A TLR2-Activating Fraction From Mycobacterium abscessus Rough Variant Demonstrates Vaccine and Diagnostic Potential.Front Cell Infect Microbiol. 2020 Aug 27;10:432. doi: 10.3389/fcimb.2020.00432. eCollection 2020. Front Cell Infect Microbiol. 2020. PMID: 32984067 Free PMC article.
-
Synergistic Efficacy of β-Lactam Combinations against Mycobacterium abscessus Pulmonary Infection in Mice.Antimicrob Agents Chemother. 2019 Jul 25;63(8):e00614-19. doi: 10.1128/AAC.00614-19. Print 2019 Aug. Antimicrob Agents Chemother. 2019. PMID: 31109979 Free PMC article.
-
Lsr2 Is an Important Determinant of Intracellular Growth and Virulence in Mycobacterium abscessus.Front Microbiol. 2019 Apr 30;10:905. doi: 10.3389/fmicb.2019.00905. eCollection 2019. Front Microbiol. 2019. PMID: 31114557 Free PMC article.
References
-
- Griffith DE, Girard WM, Wallace RJ. 1993. Clinical features of pulmonary disease caused by rapidly growing mycobacteria. An analysis of 154 patients. Am Rev Respir Dis 147:1271–1278. - PubMed
-
- Olivier KN, Weber DJ, Wallace RJ, Faiz AR, Lee J-H, Zhang Y, Brown-Elliot BA, Handler A, Wilson RW, Schechter MS, Edwards LJ, Chakraborti S, Knowles MR. 2003. Nontuberculous mycobacteria. I: multicenter prevalence study in cystic fibrosis. Am J Respir Crit Care Med 167:828–834. doi:10.1164/rccm.200207-678OC. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

