Targeted PDT agent eradicates TrkC expressing tumors via photodynamic therapy (PDT)
- PMID: 25487316
- PMCID: PMC4291778
- DOI: 10.1021/mp5005564
Targeted PDT agent eradicates TrkC expressing tumors via photodynamic therapy (PDT)
Abstract
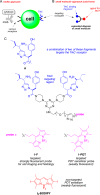
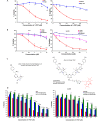
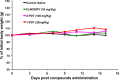
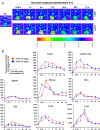
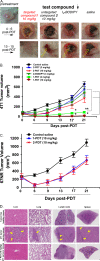
This contribution features a small molecule that binds TrkC (tropomyosin receptor kinase C) receptor that tends to be overexpressed in metastatic breast cancer cells but not in other breast cancer cells. A sensitizer for (1)O2 production conjugated to this structure gives 1-PDT for photodynamic therapy. Isomeric 2-PDT does not bind TrkC and was used as a control throughout; similarly, TrkC- cancer cells were used to calibrate enhanced killing of TrkC+ cells. Ex vivo, 1- and 2-PDT where only cytotoxic when illuminated, and 1-PDT, gave higher cell death for TrkC+ breast cancer cells. A 1 h administration-to-illumination delay gave optimal TrkC+/TrkC--photocytotoxicity, and distribution studies showed the same delay was appropriate in vivo. In Balb/c mice, a maximum tolerated dose of 20 mg/kg was determined for 1-PDT. 1- and 2-PDT (single, 2 or 10 mg/kg doses and one illumination, throughout) had similar effects on implanted TrkC- tumors, and like those of 2-PDT on TrkC+ tumors. In contrast, 1-PDT caused dramatic TrkC+ tumor volume reduction (96% from initial) relative to the TrkC- tumors or 2-PDT in TrkC+ models. Moreover, 71% of the mice treated with 10 mg/kg 1-PDT (n = 7) showed full tumor remission and survived until 90 days with no metastasis to key organs.
Keywords: histochemistry; imaging; metastatic breast cancer; photodynamic therapy (PDT); theranostic; tropomyosin receptor kinase C (TrkC).
Figures

Similar articles
-
Double-targeting using a TrkC ligand conjugated to dipyrrometheneboron difluoride (BODIPY) based photodynamic therapy (PDT) agent.J Med Chem. 2013 Oct 10;56(19):7608-14. doi: 10.1021/jm4012142. Epub 2013 Sep 24. J Med Chem. 2013. PMID: 24063347 Free PMC article.
-
Tropomyosin Receptor Kinase C Targeted Delivery of a Peptidomimetic Ligand-Photosensitizer Conjugate Induces Antitumor Immune Responses Following Photodynamic Therapy.Sci Rep. 2016 Nov 17;6:37209. doi: 10.1038/srep37209. Sci Rep. 2016. PMID: 27853305 Free PMC article.
-
Antitumor effect of 5-aminolevulinic acid-mediated photodynamic therapy can be enhanced by the use of a low dose of photofrin in human tumor xenografts.Cancer Res. 2001 Aug 1;61(15):5824-32. Cancer Res. 2001. PMID: 11479222
-
Recent strategies to improve boron dipyrromethene (BODIPY) for photodynamic cancer therapy: an updated review.Photochem Photobiol Sci. 2018 Nov 1;17(11):1691-1708. doi: 10.1039/c8pp00113h. Epub 2018 May 30. Photochem Photobiol Sci. 2018. PMID: 29845993 Review.
-
Photodynamic therapy: Inception to application in breast cancer.Breast. 2017 Feb;31:105-113. doi: 10.1016/j.breast.2016.09.016. Epub 2016 Nov 8. Breast. 2017. PMID: 27833041 Review.
Cited by
-
Click-Addressable Cassette for Photoaffinity Labeling.ACS Med Chem Lett. 2018 Jan 24;9(2):155-158. doi: 10.1021/acsmedchemlett.7b00516. eCollection 2018 Feb 8. ACS Med Chem Lett. 2018. PMID: 29456805 Free PMC article.
-
BODIPYs in PDT: A Journey through the Most Interesting Molecules Produced in the Last 10 Years.Int J Mol Sci. 2022 Sep 5;23(17):10198. doi: 10.3390/ijms231710198. Int J Mol Sci. 2022. PMID: 36077597 Free PMC article. Review.
-
A Near-IR Fluorescent Dasatinib Derivative That Localizes in Cancer Cells.Bioconjug Chem. 2019 Apr 17;30(4):1175-1181. doi: 10.1021/acs.bioconjchem.9b00118. Epub 2019 Apr 1. Bioconjug Chem. 2019. PMID: 30931563 Free PMC article.
-
One Change, Many Benefits: A Glycine-Modified Bacteriochlorin with NIR Absorption and a Type I Photochemical Mechanism for Versatile Photodynamic Therapy.Int J Mol Sci. 2024 Dec 6;25(23):13132. doi: 10.3390/ijms252313132. Int J Mol Sci. 2024. PMID: 39684841 Free PMC article.
-
Selective Modulation of Trk Receptors by Cyclo-Organopeptides.ACS Chem Neurosci. 2025 Aug 6;16(15):2776-2784. doi: 10.1021/acschemneuro.4c00833. Epub 2025 Jul 9. ACS Chem Neurosci. 2025. PMID: 40632885 Free PMC article.
References
-
- Agarwal A.; Saraf S.; Asthana A.; Gupta U.; Gajbhiye V.; Jain N. K. Ligand based dendritic systems for tumor targeting. Int. J. Pharm. 2008, 350, 3–13. - PubMed
-
- Minko T.; Dharap S. S.; Pakunlu R. I.; Wang Y. Molecular targeting of drug delivery systems to cancer. Curr. Drug Targets 2004, 5, 389–406. - PubMed
-
- Krall N.; Scheuermann J.; Neri D. Small Targeted Cytotoxics: Current State and Promises from DNA-Encoded Chemical Libraries. Angew. Chem., Int. Ed. 2013, 52, 1384–1402. - PubMed
-
- Sievers Eric L.; Senter Peter D. Antibody–drug conjugates in cancer therapy. Annu. Rev. Med. 2013, 64, 15–29. - PubMed
-
- Dennis M. S.; Jin H.; Dugger D.; Yang R.; McFarland L.; Ogasawara A.; Williams S.; Cole M. J.; Ross S.; Schwall R. Imaging Tumors with an Albumin-Binding Fab, a Novel Tumor-Targeting Agent. Cancer Res. 2007, 67, 254–261. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

