Arabidopsis receptor of activated C kinase1 phosphorylation by WITH NO LYSINE8 KINASE
- PMID: 25489024
- PMCID: PMC4326752
- DOI: 10.1104/pp.114.247460
Arabidopsis receptor of activated C kinase1 phosphorylation by WITH NO LYSINE8 KINASE
Abstract
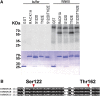
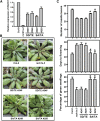
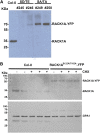
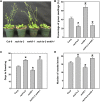
Receptor of activated C kinase1 (RACK1) is a versatile scaffold protein that binds to numerous proteins to regulate diverse cellular pathways in mammals. In Arabidopsis (Arabidopsis thaliana), RACK1 has been shown to regulate plant hormone signaling, stress responses, and multiple processes of growth and development. However, little is known about the molecular mechanism underlying these regulations. Here, we show that an atypical serine (Ser)/threonine (Thr) protein kinase, WITH NO LYSINE8 (WNK8), phosphorylates RACK1. WNK8 physically interacted with and phosphorylated RACK1 proteins at two residues: Ser-122 and Thr-162. Genetic epistasis analysis of rack1 wnk8 double mutants indicated that RACK1 acts downstream of WNK8 in the glucose responsiveness and flowering pathways. The phosphorylation-dead form, RACK1A(S122A/T162A), but not the phosphomimetic form, RACK1A(S122D/T162E), rescued the rack1a null mutant, implying that phosphorylation at Ser-122 and Thr-162 negatively regulates RACK1A function. The transcript of RACK1A(S122D/T162E) accumulated at similar levels as those of RACK1(S122A/T162A). However, although the steady-state level of the RACK1A(S122A/T162A) protein was similar to wild-type RACK1A protein, the RACK1A(S122D/T162E) protein was nearly undetectable, suggesting that phosphorylation affects the stability of RACK1A proteins. Taken together, these results suggest that RACK1 is phosphorylated by WNK8 and that phosphorylation negatively regulates RACK1 function by influencing its protein stability.
© 2015 American Society of Plant Biologists. All Rights Reserved.
Figures



Similar articles
-
Phosphorylation of RACK1 in plants.Plant Signal Behav. 2015;10(8):e1022013. doi: 10.1080/15592324.2015.1022013. Plant Signal Behav. 2015. PMID: 26322575 Free PMC article. Review.
-
RACK1 genes regulate plant development with unequal genetic redundancy in Arabidopsis.BMC Plant Biol. 2008 Oct 23;8:108. doi: 10.1186/1471-2229-8-108. BMC Plant Biol. 2008. PMID: 18947417 Free PMC article.
-
Dissection of the relationship between RACK1 and heterotrimeric G-proteins in Arabidopsis.Plant Cell Physiol. 2009 Sep;50(9):1681-94. doi: 10.1093/pcp/pcp113. Epub 2009 Aug 3. Plant Cell Physiol. 2009. PMID: 19651700
-
RACK1 is a negative regulator of ABA responses in Arabidopsis.J Exp Bot. 2009;60(13):3819-33. doi: 10.1093/jxb/erp221. Epub 2009 Jul 7. J Exp Bot. 2009. PMID: 19584117 Free PMC article.
-
The Receptor for Activated C Kinase in Plant Signaling: Tale of a Promiscuous Little Molecule.Front Plant Sci. 2015 Dec 8;6:1090. doi: 10.3389/fpls.2015.01090. eCollection 2015. Front Plant Sci. 2015. PMID: 26697044 Free PMC article. Review.
Cited by
-
Broad Substrate-Specific Phosphorylation Events Are Associated With the Initial Stage of Plant Cell Wall Recognition in Neurospora crassa.Front Microbiol. 2019 Nov 1;10:2317. doi: 10.3389/fmicb.2019.02317. eCollection 2019. Front Microbiol. 2019. PMID: 31736884 Free PMC article.
-
Alteration of cell wall xylan acetylation triggers defense responses that counterbalance the immune deficiencies of plants impaired in the β-subunit of the heterotrimeric G-protein.Plant J. 2017 Nov;92(3):386-399. doi: 10.1111/tpj.13660. Epub 2017 Sep 15. Plant J. 2017. PMID: 28792629 Free PMC article.
-
Genome-Wide Identification and Analysis of the WNK Kinase Gene Family in Upland Cotton.Plants (Basel). 2023 Nov 30;12(23):4036. doi: 10.3390/plants12234036. Plants (Basel). 2023. PMID: 38068671 Free PMC article.
-
Tyrosine Phosphorylation Based Homo-dimerization of Arabidopsis RACK1A Proteins Regulates Oxidative Stress Signaling Pathways in Yeast.Front Plant Sci. 2016 Feb 24;7:176. doi: 10.3389/fpls.2016.00176. eCollection 2016. Front Plant Sci. 2016. PMID: 26941753 Free PMC article.
-
Phosphorylation of RACK1 in plants.Plant Signal Behav. 2015;10(8):e1022013. doi: 10.1080/15592324.2015.1022013. Plant Signal Behav. 2015. PMID: 26322575 Free PMC article. Review.
References
-
- Chang BY, Harte RA, Cartwright CA (2002) RACK1: a novel substrate for the Src protein-tyrosine kinase. Oncogene 21: 7619–7629 - PubMed
-
- Chen JG, Ullah H, Temple B, Liang J, Guo J, Alonso JM, Ecker JR, Jones AM (2006b) RACK1 mediates multiple hormone responsiveness and developmental processes in Arabidopsis. J Exp Bot 57: 2697–2708 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

