αCGRP is essential for algesic exocytotic mobilization of TRPV1 channels in peptidergic nociceptors
- PMID: 25489075
- PMCID: PMC4280602
- DOI: 10.1073/pnas.1420252111
αCGRP is essential for algesic exocytotic mobilization of TRPV1 channels in peptidergic nociceptors
Abstract
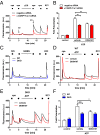
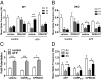
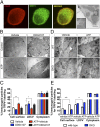
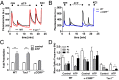
Proalgesic sensitization of peripheral nociceptors in painful syndromes is a complex molecular process poorly understood that involves mobilization of thermosensory receptors to the neuronal surface. However, whether recruitment of vesicular thermoTRP channels is a general mechanism underlying sensitization of all nociceptor types or is subtype-specific remains controversial. We report that sensitization-induced Ca(2+)-dependent exocytotic insertion of transient receptor potential vanilloid 1 (TRPV1) receptors to the neuronal plasma membrane is a mechanism specifically used by peptidergic nociceptors to potentiate their excitability. Notably, we found that TRPV1 is present in large dense-core vesicles (LDCVs) that were mobilized to the neuronal surface in response to a sensitizing insult. Deletion or silencing of calcitonin-gene-related peptide alpha (αCGRP) gene expression drastically reduced proalgesic TRPV1 potentiation in peptidergic nociceptors by abrogating its Ca(2+)-dependent exocytotic recruitment. These findings uncover a context-dependent molecular mechanism of TRPV1 algesic sensitization and a previously unrecognized role of αCGRP in LDCV mobilization in peptidergic nociceptors. Furthermore, these results imply that concurrent secretion of neuropeptides and channels in peptidergic C-type nociceptors facilitates a rapid modulation of pain signaling.
Keywords: inflammation; ion channel; nociception; pain transduction; sensory neuron.
Conflict of interest statement
Conflict of interest statement: A.F.-M. is an inventor of patent WO2010 009892, protecting the antinociceptive activity of compound DD04107.
Figures


Similar articles
-
Bradykinin Induces TRPV1 Exocytotic Recruitment in Peptidergic Nociceptors.Front Pharmacol. 2016 Jun 23;7:178. doi: 10.3389/fphar.2016.00178. eCollection 2016. Front Pharmacol. 2016. PMID: 27445816 Free PMC article.
-
Differential contribution of SNARE-dependent exocytosis to inflammatory potentiation of TRPV1 in nociceptors.FASEB J. 2009 Nov;23(11):3722-33. doi: 10.1096/fj.09-134346. Epub 2009 Jul 7. FASEB J. 2009. PMID: 19584302
-
Increased expression of Trpv1 in peripheral terminals mediates thermal nociception in Fabry disease mouse model.Mol Pain. 2016 Aug 16;12:1744806916663729. doi: 10.1177/1744806916663729. Print 2016. Mol Pain. 2016. PMID: 27531673 Free PMC article.
-
The mechanism of μ-opioid receptor (MOR)-TRPV1 crosstalk in TRPV1 activation involves morphine anti-nociception, tolerance and dependence.Channels (Austin). 2015;9(5):235-43. doi: 10.1080/19336950.2015.1069450. Epub 2015 Jul 15. Channels (Austin). 2015. PMID: 26176938 Free PMC article. Review.
-
Functional aspects and mechanisms of TRPV1 involvement in neurogenic inflammation that leads to thermal hyperalgesia.Pflugers Arch. 2005 Oct;451(1):151-9. doi: 10.1007/s00424-005-1423-5. Epub 2005 May 21. Pflugers Arch. 2005. PMID: 15909179 Review.
Cited by
-
CGRP in Animal Models of Migraine.Handb Exp Pharmacol. 2019;255:85-107. doi: 10.1007/164_2018_187. Handb Exp Pharmacol. 2019. PMID: 30689086 Free PMC article. Review.
-
TNFα induces co-trafficking of TRPV1/TRPA1 in VAMP1-containing vesicles to the plasmalemma via Munc18-1/syntaxin1/SNAP-25 mediated fusion.Sci Rep. 2016 Feb 18;6:21226. doi: 10.1038/srep21226. Sci Rep. 2016. PMID: 26888187 Free PMC article.
-
NGF Enhances CGRP Release Evoked by Capsaicin from Rat Trigeminal Neurons: Differential Inhibition by SNAP-25-Cleaving Proteases.Int J Mol Sci. 2022 Jan 14;23(2):892. doi: 10.3390/ijms23020892. Int J Mol Sci. 2022. PMID: 35055082 Free PMC article.
-
The potentiating effect of calcitonin gene-related peptide on transient receptor potential vanilloid-1 activity and the electrophysiological responses of rat trigeminal neurons to nociceptive stimuli.J Physiol Sci. 2018 May;68(3):261-268. doi: 10.1007/s12576-017-0529-9. Epub 2017 Feb 15. J Physiol Sci. 2018. PMID: 28205139 Free PMC article.
-
Mechanism of Action of OnabotulinumtoxinA in Chronic Migraine: A Narrative Review.Headache. 2020 Jul;60(7):1259-1272. doi: 10.1111/head.13849. Epub 2020 Jun 30. Headache. 2020. PMID: 32602955 Free PMC article. Review.
References
-
- Caterina MJ, Julius D. The vanilloid receptor: A molecular gateway to the pain pathway. Annu Rev Neurosci. 2001;24:487–517. - PubMed
-
- Kim HY, et al. Differential changes in TRPV1 expression after trigeminal sensory nerve injury. J Pain. 2008;9(3):280–288. - PubMed
-
- Pabbidi RM, Cao DS, Parihar A, Pauza ME, Premkumar LS. Direct role of streptozotocin in inducing thermal hyperalgesia by enhanced expression of transient receptor potential vanilloid 1 in sensory neurons. Mol Pharmacol. 2008;73(3):995–1004. - PubMed
-
- Szabó A, et al. Role of transient receptor potential vanilloid 1 receptors in adjuvant-induced chronic arthritis: In vivo study using gene-deficient mice. J Pharmacol Exp Ther. 2005;314(1):111–119. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

