Plasma and cerebrospinal fluid herpes simplex virus levels at diagnosis and outcome of neonatal infection
- PMID: 25491092
- PMCID: PMC4380781
- DOI: 10.1016/j.jpeds.2014.11.011
Plasma and cerebrospinal fluid herpes simplex virus levels at diagnosis and outcome of neonatal infection
Abstract
Objective: To evaluate the utility of quantitative herpes simplex virus (HSV) polymerase chain reaction (PCR) levels for prognosis and management of neonatal HSV disease.
Study design: Clinical and virologic data were abstracted by medical record review from neonatal HSV cases treated at Seattle Children's Hospital between 1993 and 2012. HSV PCR results from plasma (n = 47), cerebrospinal fluid (n = 56), or both (n = 40) at the time of diagnosis were available from 63 infants; 26 with skin-eye-mouth (SEM), 18 with central nervous system (CNS), and 19 with disseminated (DIS) disease.
Results: Plasma HSV PCR was positive in 78% of the infants with SEM, 64% with CNS and 100% with DIS disease. Mean plasma viral level was 2.8 log10 copies/mL in SEM, 2.2 log10 copies/mL in CNS, and 7.2 log10 copies/mL in DIS infants. The HSV levels were higher among infants who died compared with surviving infants, 8.1 log10 copies/mL (range 7.7-8.6) vs 3.8 log10 copies/mL (range 0.0-8.6), P = .001, however, level of HSV DNA in the cerebrospinal fluid or in plasma did not correlate with neurologic outcome. Dynamics of HSV clearance from plasma during high-dose acyclovir treatment showed single-phase exponential decay with a median viral half-life of 1.26 days (range: 0.8-1.51).
Conclusions: Plasma HSV levels correlate with clinical presentation of neonatal HSV disease and mortality, but not neurologic outcome.
Copyright © 2015 Elsevier Inc. All rights reserved.
Conflict of interest statement
The other authors declare no conflicts of interest.
Figures
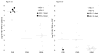

Comment in
-
Quantitative herpes simplex virus concentrations in neonatal infection.J Pediatr. 2015 Apr;166(4):793-5. doi: 10.1016/j.jpeds.2014.12.077. Epub 2015 Feb 7. J Pediatr. 2015. PMID: 25662831 No abstract available.
Similar articles
-
Utility of Surface and Blood Polymerase Chain Reaction Assays in Identifying Infants With Neonatal Herpes Simplex Virus Infection.Pediatr Infect Dis J. 2019 Nov;38(11):1138-1140. doi: 10.1097/INF.0000000000002451. Pediatr Infect Dis J. 2019. PMID: 31626049
-
Application of the polymerase chain reaction to the diagnosis and management of neonatal herpes simplex virus disease. National Institute of Allergy and Infectious Diseases Collaborative Antiviral Study Group.J Infect Dis. 1996 Dec;174(6):1162-7. doi: 10.1093/infdis/174.6.1162. J Infect Dis. 1996. PMID: 8940204
-
Diagnostic and therapeutic management for suspected neonatal herpes simplex virus infection.J Clin Virol. 2011 May;51(1):8-11. doi: 10.1016/j.jcv.2011.02.008. Epub 2011 Mar 8. J Clin Virol. 2011. PMID: 21388869
-
Herpes simplex virus, meningitis and encephalitis in neonates.Herpes. 2004 Jun;11 Suppl 2:65A-76A. Herpes. 2004. PMID: 15319092 Review.
-
Management of neonatal herpes simplex virus infection.Paediatr Drugs. 2001;3(2):81-90. doi: 10.2165/00128072-200103020-00001. Paediatr Drugs. 2001. PMID: 11269641 Review.
Cited by
-
Treatment of perinatal viral infections to improve neurologic outcomes.Pediatr Res. 2017 Jan;81(1-2):162-169. doi: 10.1038/pr.2016.191. Epub 2016 Sep 27. Pediatr Res. 2017. PMID: 27673425 Review.
-
Predominant area of brain lesions in neonates with herpes simplex encephalitis.J Perinatol. 2017 Nov;37(11):1210-1214. doi: 10.1038/jp.2017.114. Epub 2017 Jul 20. J Perinatol. 2017. PMID: 28726789
-
Trivalent Glycoprotein Subunit Vaccine Prevents Neonatal Herpes Simplex Virus Mortality and Morbidity.J Virol. 2020 May 18;94(11):e02163-19. doi: 10.1128/JVI.02163-19. Print 2020 May 18. J Virol. 2020. PMID: 32188735 Free PMC article.
-
Epidemiology and treatment of herpes simplex virus in the neonatal intensive care unit.J Perinatol. 2025 Jan;45(1):116-121. doi: 10.1038/s41372-024-02150-8. Epub 2024 Oct 11. J Perinatol. 2025. PMID: 39394454
-
Neonatal Herpes Simplex Viral Infections and Acyclovir: An Update.J Pediatr Pharmacol Ther. 2017 Mar-Apr;22(2):88-93. doi: 10.5863/1551-6776-22.2.88. J Pediatr Pharmacol Ther. 2017. PMID: 28469532 Free PMC article. Review.
References
-
- Caviness AC, Demmler GJ, Swint JM, Cantor SB. Cost-effectiveness analysis of herpes simplex virus testing and treatment strategies in febrile neonates. Arch Pediatr Adolesc Med. 2008;162:665–74. - PubMed
-
- Kimberlin DW. Diagnosis of herpes simplex virus in the era of polymerase chain reaction. Pediatr Infect Dis J. 2006;25:841–2. - PubMed
-
- Cantey JB, Mejias A, Wallihan R, Doern C, Brock E, Salamon D, et al. Use of blood polymerase chain reaction testing for diagnosis of herpes simplex virus infection. J Pediatr. 2012;161:357–61. - PubMed
-
- Kimberlin DW, Lakeman FD, Arvin AM, Prober CG, Corey L, Powell DA, et al. Application of the polymerase chain reaction to the diagnosis and management of neonatal herpes simplex virus disease. National Institute of Allergy and Infectious Diseases Collaborative Antiviral Study Group. J Infect Dis. 1996;174:1162–7. - PubMed
Publication types
MeSH terms
Substances
Supplementary concepts
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

