Integrins in Wound Healing
- PMID: 25493210
- PMCID: PMC4250945
- DOI: 10.1089/wound.2013.0436
Integrins in Wound Healing
Abstract
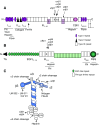
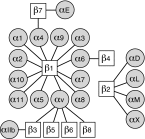
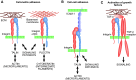
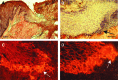
Significance: Regulation of cell adhesions during tissue repair is fundamentally important for cell migration, proliferation, and protein production. All cells interact with extracellular matrix proteins with cell surface integrin receptors that convey signals from the environment into the nucleus, regulating gene expression and cell behavior. Integrins also interact with a variety of other proteins, such as growth factors, their receptors, and proteolytic enzymes. Re-epithelialization and granulation tissue formation are crucially dependent on the temporospatial function of multiple integrins. This review explains how integrins function in wound repair. Recent Advances: Certain integrins can activate latent transforming growth factor beta-1 (TGF-β1) that modulates wound inflammation and granulation tissue formation. Dysregulation of TGF-β1 function is associated with scarring and fibrotic disorders. Therefore, these integrins represent targets for therapeutic intervention in fibrosis. Critical Issues: Integrins have multifaceted functions and extensive crosstalk with other cell surface receptors and molecules. Moreover, in aberrant healing, integrins may assume different functions, further increasing the complexity of their functionality. Discovering and understanding the role that integrins play in wound healing provides an opportunity to identify the mechanisms for medical conditions, such as excessive scarring, chronic wounds, and even cancer. Future Directions: Integrin functions in acute and chronic wounds should be further addressed in models better mimicking human wounds. Application of any products in acute or chronic wounds will potentially alter integrin functions that need to be carefully considered in the design.
Figures






References
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

