Laser-induced porous graphene films from commercial polymers
- PMID: 25493446
- PMCID: PMC4264682
- DOI: 10.1038/ncomms6714
Laser-induced porous graphene films from commercial polymers
Abstract
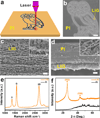
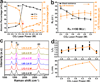
The cost effective synthesis and patterning of carbon nanomaterials is a challenge in electronic and energy storage devices. Here we report a one-step, scalable approach for producing and patterning porous graphene films with three-dimensional networks from commercial polymer films using a CO2 infrared laser. The sp3-carbon atoms are photothermally converted to sp2-carbon atoms by pulsed laser irradiation. The resulting laser-induced graphene (LIG) exhibits high electrical conductivity. The LIG can be readily patterned to interdigitated electrodes for in-plane microsupercapacitors with specific capacitances of >4 mF cm-2 and power densities of ~9 mW cm-2. Theoretical calculations partially suggest that enhanced capacitance may result from LIG's unusual ultra-polycrystalline lattice of pentagon-heptagon structures. Combined with the advantage of one-step processing of LIG in air from commercial polymer sheets, which would allow the employment of a roll-to-roll manufacturing process, this technique provides a rapid route to polymer-written electronic and energy storage devices.
Figures


References
-
- Geim AK. Graphene: status and prospects. Science. 2009;324:1530–1534. - PubMed
-
- Chen ZP, et al. Three-dimensional flexible and conductive interconnected graphene networks grown by chemical vapour deposition. Nat. Mater. 2011;10:424–428. - PubMed
-
- Miller JR, Outlaw RA, Holloway BC. Graphene double-layer capacitor with ac line-filtering performance. Science. 2010;329:1637–1639. - PubMed
-
- Wu ZS, et al. Three-dimensional nitrogen and boron co-doped graphene for high-performance all-solid-state supercapacitors. Adv. Mater. 2012;24:5130–5135. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

