Identification of Dipeptidyl-Peptidase (DPP)5 and DPP7 in Porphyromonas endodontalis, Distinct from Those in Porphyromonas gingivalis
- PMID: 25494328
- PMCID: PMC4262410
- DOI: 10.1371/journal.pone.0114221
Identification of Dipeptidyl-Peptidase (DPP)5 and DPP7 in Porphyromonas endodontalis, Distinct from Those in Porphyromonas gingivalis
Abstract
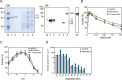
Dipeptidyl peptidases (DPPs) that liberate dipeptides from the N-terminal end of oligopeptides are crucial for the growth of Porphyromonas species, anaerobic asaccharolytic gram negative rods that utilize amino acids as energy sources. Porphyromonas endodontalis is a causative agent of periapical lesions with acute symptoms and Asp/Glu-specific DPP11 has been solely characterized in this organism. In this study, we identified and characterized two P. endodontalis DPPs, DPP5 and DPP7. Cell-associated DPP activity toward Lys-Ala-4-methylcoumaryl-7-amide (MCA) was prominent in P. endodontalis ATCC 35406 as compared with the Porphyromonas gingivalis strains ATCC 33277, 16-1, HW24D1, ATCC 49417, W83, W50, and HNA99. The level of hydrolysis of Leu-Asp-MCA by DPP11, Gly-Pro-MCA by DPP4, and Met-Leu-MCA was also higher than in the P. gingivalis strains. MER236725 and MER278904 are P. endodontalis proteins belong to the S9- and S46-family peptidases, respectively. Recombinant MER236725 exhibited enzymatic properties including substrate specificity, and salt- and pH-dependence similar to P. gingivalis DPP5 belonging to the S9 family. However, the kcat/Km figure (194 µM-1·sec-1) for the most potent substrate (Lys-Ala-MCA) was 18.4-fold higher as compared to the P. gingivalis entity (10.5 µM-1·sec-1). In addition, P. endodontalis DPP5 mRNA and protein contents were increased several fold as compared with those in P. gingivalis. Recombinant MER278904 preferentially hydrolyzed Met-Leu-MCA and exhibited a substrate specificity similar to P. gingivalis DPP7 belonging to the S46 family. In accord with the deduced molecular mass of 818 amino acids, a 105-kDa band was immunologically detected, indicating that P. endodontalis DPP7 is an exceptionally large molecule in the DPP7/DPP11/S46 peptidase family. The enhancement of four DPP activities was conclusively demonstrated in P. endodontalis, and remarkable Lys-Ala-MCA-hydrolysis was achieved by qualitative and quantitative potentiation of the DPP5 molecule.
Conflict of interest statement
Figures





References
-
- van Steenbergen TJM, Van Winkelhoff AJ, Mayrand D, Grenier D, De Graaff J (1984) Bacteroides endodontalis sp. nov., an asaccharolytic black-pigmented Bacteroides species from infected dental root canals. Int J Syst Bacteriol 34:118–120.
-
- Sundqvist G, Johansson E, Sjögren U (1989) Prevalence of black-pigmented bacteroides species in root canal infections. J Endod 15:13–19. - PubMed
-
- Flynn TR, Paster BJ, Stokes LN, Susarla SM, Shanti RM (2012) Molecular methods for diagnosis of odontogenic infections. J Oral Maxillofac Surg 70:1854–1859. - PubMed
-
- Citron DM, Poxton IR, Jo Baron E (2007) Bacteroides, Porphyromonas, Prevotella, Fusobacterium, and other anaerobic gram-negative rods. In:Murray PR, Jo Baron E, Landry, ML, Jorgensen JH, Pfaller MAeditors. Manual of Clinical Microbiology. Washington, DC: ASM Press. pp. 911–932.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

