Naturally selected hepatitis C virus polymorphisms confer broad neutralizing antibody resistance
- PMID: 25500884
- PMCID: PMC4382262
- DOI: 10.1172/JCI78794
Naturally selected hepatitis C virus polymorphisms confer broad neutralizing antibody resistance
Abstract
For hepatitis C virus (HCV) and other highly variable viruses, broadly neutralizing mAbs are an important guide for vaccine development. The development of resistance to anti-HCV mAbs is poorly understood, in part due to a lack of neutralization testing against diverse, representative panels of HCV variants. Here, we developed a neutralization panel expressing diverse, naturally occurring HCV envelopes (E1E2s) and used this panel to characterize neutralizing breadth and resistance mechanisms of 18 previously described broadly neutralizing anti-HCV human mAbs. The observed mAb resistance could not be attributed to polymorphisms in E1E2 at known mAb-binding residues. Additionally, hierarchical clustering analysis of neutralization resistance patterns revealed relationships between mAbs that were not predicted by prior epitope mapping, identifying 3 distinct neutralization clusters. Using this clustering analysis and envelope sequence data, we identified polymorphisms in E2 that confer resistance to multiple broadly neutralizing mAbs. These polymorphisms, which are not at mAb contact residues, also conferred resistance to neutralization by plasma from HCV-infected subjects. Together, our method of neutralization clustering with sequence analysis reveals that polymorphisms at noncontact residues may be a major immune evasion mechanism for HCV, facilitating viral persistence and presenting a challenge for HCV vaccine development.
Figures

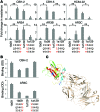





Comment in
-
The genetics of hepatitis C virus underlie its ability to escape humoral immunity.J Clin Invest. 2015 Jan;125(1):97-8. doi: 10.1172/JCI79424. Epub 2014 Dec 15. J Clin Invest. 2015. PMID: 25500881 Free PMC article.
References
-
- Lawitz E, et al. Sofosbuvir in combination with peginterferon alfa-2a and ribavirin for non-cirrhotic, treatment-naive patients with genotypes 1, 2, and 3 hepatitis C infection: a randomised, double-blind, phase 2 trial. Lancet Infect Dis. 2013;13(5):401–408. doi: 10.1016/S1473-3099(13)70033-1. - DOI - PubMed
-
- Liu L, Fisher BE, Dowd KA, Astemborski J, Cox AL, Ray SC. Acceleration of hepatitis C virus envelope evolution in humans is consistent with progressive humoral immune selection during the transition from acute to chronic infection. J Virol. 2010;84(10):5067–5077. doi: 10.1128/JVI.02265-09. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

