p38α (MAPK14) critically regulates the immunological response and the production of specific cytokines and chemokines in astrocytes
- PMID: 25502009
- PMCID: PMC4264013
- DOI: 10.1038/srep07405
p38α (MAPK14) critically regulates the immunological response and the production of specific cytokines and chemokines in astrocytes
Abstract
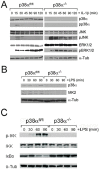
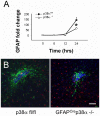
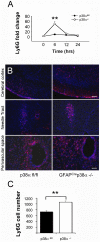
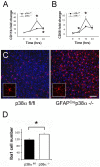
In CNS lesions, "reactive astrocytes" form a prominent cellular response. However, the nature of this astrocyte immune activity is not well understood. In order to study astrocytic immune responses to inflammation and injury, we generated mice with conditional deletion of p38α (MAPK14) in GFAP+ astrocytes. We studied the role of p38α signaling in astrocyte immune activation both in vitro and in vivo, and simultaneously examined the effects of astrocyte activation in CNS inflammation. Our results showed that specific subsets of cytokines (TNFα, IL-6) and chemokines (CCL2, CCL4, CXCL1, CXCL2, CXCL10) are critically regulated by p38α signaling in astrocytes. In an in vivo CNS inflammation model of intracerebral injection of LPS, we observed markedly attenuated astrogliosis in conditional GFAPcre p38α(-/-) mice. However, GFAPcre p38α(-/-) mice showed marked upregulation of CCL2, CCL3, CCL4, CXCL2, CXCL10, TNFα, and IL-1β compared to p38αfl/fl cohorts, suggesting that in vivo responses to LPS after GFAPcre p38α deletion are complex and involve interactions between multiple cell types. This finding was supported by a prominent increase in macrophage/microglia and neutrophil recruitment in GFAPcre p38α(-/-) mice compared to p38αfl/fl controls. Together, these studies provide important insights into the critical role of p38α signaling in astrocyte immune activation.
Figures






Similar articles
-
IL-1β induces GFAP expression in vitro and in vivo and protects neurons from traumatic injury-associated apoptosis in rat brain striatum via NFκB/Ca²⁺-calmodulin/ERK mitogen-activated protein kinase signaling pathway.Neuroscience. 2013 Nov 12;252:367-83. doi: 10.1016/j.neuroscience.2013.07.061. Epub 2013 Aug 6. Neuroscience. 2013. PMID: 23928073
-
Redox regulation of glial inflammatory response to lipopolysaccharide and interferongamma.J Neurosci Res. 2004 Aug 15;77(4):540-51. doi: 10.1002/jnr.20180. J Neurosci Res. 2004. PMID: 15264224
-
Control of IL-17 receptor signaling and tissue inflammation by the p38α-MKP-1 signaling axis in a mouse model of multiple sclerosis.Sci Signal. 2015 Mar 3;8(366):ra24. doi: 10.1126/scisignal.aaa2147. Sci Signal. 2015. PMID: 25737586 Free PMC article.
-
The opioid antagonist, β-funaltrexamine, inhibits NF-κB signaling and chemokine expression in human astrocytes and in mice.Eur J Pharmacol. 2015 Sep 5;762:193-201. doi: 10.1016/j.ejphar.2015.05.040. Epub 2015 May 22. Eur J Pharmacol. 2015. PMID: 26007645 Free PMC article.
-
Astrocyte-immune cell interactions in physiology and pathology.Immunity. 2021 Feb 9;54(2):211-224. doi: 10.1016/j.immuni.2021.01.013. Immunity. 2021. PMID: 33567261 Review.
Cited by
-
Predicting Clinical Dementia Rating Using Blood RNA Levels.Genes (Basel). 2020 Jun 26;11(6):706. doi: 10.3390/genes11060706. Genes (Basel). 2020. PMID: 32604772 Free PMC article.
-
The role of senescence-related genes in major depressive disorder: insights from machine learning and single cell analysis.BMC Psychiatry. 2025 Mar 3;25(1):188. doi: 10.1186/s12888-025-06542-8. BMC Psychiatry. 2025. PMID: 40033248 Free PMC article.
-
SIRT2 Plays Significant Roles in Lipopolysaccharides-Induced Neuroinflammation and Brain Injury in Mice.Neurochem Res. 2016 Sep;41(9):2490-500. doi: 10.1007/s11064-016-1981-2. Epub 2016 Jun 27. Neurochem Res. 2016. PMID: 27350577
-
Mapping p38α mitogen-activated protein kinase signaling by proximity-dependent labeling.Protein Sci. 2020 May;29(5):1196-1210. doi: 10.1002/pro.3854. Epub 2020 Apr 7. Protein Sci. 2020. PMID: 32189389 Free PMC article.
-
A comparative transcriptomic analysis of astrocytes differentiation from human neural progenitor cells.Eur J Neurosci. 2016 Nov;44(10):2858-2870. doi: 10.1111/ejn.13382. Epub 2016 Sep 25. Eur J Neurosci. 2016. PMID: 27564458 Free PMC article.
References
-
- Bergles D. E., Diamond J. S. & Jahr C. E. Clearance of glutamate inside the synapse and beyond. Curr Opin Neurobiol 9, 293–298 (1999). - PubMed
-
- Nagelhus E. A. et al. Immunogold evidence suggests that coupling of K+ siphoning and water transport in rat retinal Muller cells is mediated by a coenrichment of Kir4.1 and AQP4 in specific membrane domains. Glia 26, 47–54 (1999). - PubMed
-
- Brown A. M. & Ransom B. R. Astrocyte glycogen and brain energy metabolism. Glia 55, 1263–1271 (2007). - PubMed
-
- Piet R., Poulain D. A. & Oliet S. H. Contribution of astrocytes to synaptic transmission in the rat supraoptic nucleus. Neurochem Int 45, 251–257 (2004). - PubMed
-
- Abbott N. J., Ronnback L. & Hansson E. Astrocyte-endothelial interactions at the blood-brain barrier. Nat Rev Neurosci 7, 41–53 (2006). - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

