A peculiar mutation spectrum emerging from young peruvian patients with hepatocellular carcinoma
- PMID: 25502816
- PMCID: PMC4263719
- DOI: 10.1371/journal.pone.0114912
A peculiar mutation spectrum emerging from young peruvian patients with hepatocellular carcinoma
Abstract
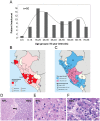
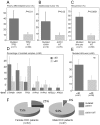
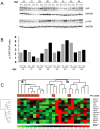
Hepatocellular carcinoma usually afflicts individuals in their later years following longstanding liver disease. In Peru, hepatocellular carcinoma exists in a unique clinical presentation, which affects patients around age 25 with a normal, healthy liver. In order to deepen our understanding of the molecular processes ongoing in Peruvian liver tumors, mutation spectrum analysis was carried out on hepatocellular carcinomas from 80 Peruvian patients. Sequencing analysis focused on nine genes typically altered during liver carcinogenesis, i.e. ARID2, AXIN1, BRAF, CTNNB1, NFE2L2, H/K/N-RAS, and TP53. We also assessed the transcription level of factors involved in the control of the alpha-fetoprotein expression and the Hippo signaling pathway that controls contact inhibition in metazoans. The mutation spectrum of Peruvian patients was unique with a major class of alterations represented by Insertions/Deletions. There were no changes at hepatocellular carcinoma-associated mutation hotspots in more than half of the specimens analyzed. Furthermore, our findings support the theory of a consistent collapse in the Hippo axis, as well as an expression of the stemness factor NANOG in high alpha-fetoprotein-expressing hepatocellular carcinomas. These results confirm the specificity of Peruvian hepatocellular carcinoma at the molecular genetic level. The present study emphasizes the necessity to widen cancer research to include historically neglected patients from South America, and more broadly the Global South, where cancer genetics and tumor presentation are divergent from canonical neoplasms.
Conflict of interest statement
Figures



References
-
- Ferlay J, Soerjomataram I, Dikshit R, Eser S, Mathers C, et al. (2014) Cancer incidence and mortality worldwide: Sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer doi:10.1002/ijc.29210. - DOI - PubMed
-
- McGlynn K, London W (2005) Epidemiology and natural history of hepatocellular carcinoma. Best Pract Res Clin Gastroenterol 19:3–23. - PubMed
-
- Goss PE, Lee BL, Badonivac-Crnjevic T, Strasser-Weippl K, Chavarri-Guerra Y, et al. (2013) Planning cancer control in Latin America and the Caribbean. Lancet Oncol 14:391–436. - PubMed
-
- Ruiz E, Sanchez J, Celis J, Payet E, Berrospi F, et al. (2007) [Short and long-term results of liver resection for hepatocellular carcinoma in Peru: a Peruvian single center experience on 232 cases.]. Rev Gastoenterol Peru 27:223–237 Spanish - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

