Intracellular signaling mechanisms of the melanocortin receptors: current state of the art
- PMID: 25504085
- PMCID: PMC11113477
- DOI: 10.1007/s00018-014-1800-3
Intracellular signaling mechanisms of the melanocortin receptors: current state of the art
Abstract
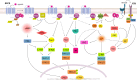
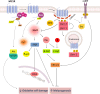
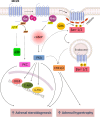
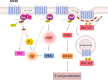
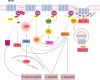
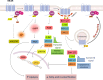
The melanocortin system is composed by the agonists adrenocorticotropic hormone and α, β and γ-melanocyte-stimulating hormone, and two naturally occurring antagonists, agouti and agouti-related protein. These ligands act by interaction with a family of five melanocortin receptors (MCRs), assisted by MCRs accessory proteins (MRAPs). MCRs stimulation activates different signaling pathways that mediate a diverse array of physiological processes, including pigmentation, energy metabolism, inflammation and exocrine secretion. This review focuses on the regulatory mechanisms of MCRs signaling, highlighting the differences among the five receptors. MCRs signal through G-dependent and independent mechanisms and their functional coupling to agonists at the cell surface is regulated by interacting proteins, namely MRAPs and β-arrestins. The knowledge of the distinct modulation pattern of MCRs signaling and function may be helpful for the future design of novel drugs able to combine specificity, safety and effectiveness in the course of their therapeutic use.
Conflict of interest statement
The authors have no conflict of interest.
Figures
References
-
- Cooray SN, Clark AJ. Melanocortin receptors and their accessory proteins. Mol Cell Endocrinol. 2011;331:215–221. - PubMed
-
- Smith AI, Funder JW Proopiomelanocortin processing in the pituitary, central nervous system, and peripheral tissues. Endocr Rev. 1988;9:159–179. - PubMed
-
- Bultman SJ, Michaud EJ, Woychik RP. Molecular characterization of the mouse agouti locus. Cell. 1992;71:1195–1204. - PubMed
-
- Wilson BD, Ollmann MM, Kang L, Stoffel M, Bell GI, Barsh GS. Structure and function of ASP, the human homolog of the mouse agouti gene. Hum Mol Genet. 1995;4:223–230. - PubMed
-
- Voisey J, Kelly G, Van Daal A. Agouti signal protein regulation in human melanoma cells. Pigment Cell Res. 2003;16:65–71. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

