Purine receptors are required for DHA-mediated neuroprotection against oxygen and glucose deprivation in hippocampal slices
- PMID: 25504554
- PMCID: PMC4336303
- DOI: 10.1007/s11302-014-9438-z
Purine receptors are required for DHA-mediated neuroprotection against oxygen and glucose deprivation in hippocampal slices
Abstract
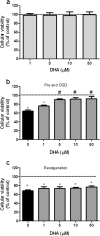
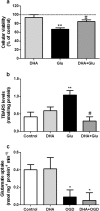
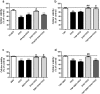
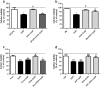
Docosahexaenoic acid (DHA) is important for central nervous system function during pathological states such as ischemia. DHA reduces neuronal injury in experimental brain ischemia; however, the underlying mechanisms are not well understood. In the present study, we investigated the effects of DHA on acute hippocampal slices subjected to experimental ischemia by transient oxygen and glucose deprivation (OGD) and re-oxygenation and the possible involvement of purinergic receptors as the mechanism underlying DHA-mediated neuroprotection. We observed that cellular viability reduction induced by experimental ischemia as well as cell damage and thiobarbituric acid reactive substances (TBARS) production induced by glutamate (10 mM) were prevented by hippocampal slices pretreated with DHA (5 μM). However, glutamate uptake reduction induced by OGD and re-oxygenation was not prevented by DHA. The beneficial effect of DHA against cellular viability reduction induced by OGD and re-oxygenation was blocked with PPADS (3 μM), a nonselective P2X1-5 receptor antagonist as well as with a combination of TNP-APT (100 nM) plus brilliant blue (100 nM), which blocked P2X1, P2X3, P2X2/3, and P2X7 receptors, respectively. Moreover, adenosine receptors blockade with A1 receptor antagonist DPCPX (100 nM) or with A2B receptor antagonist alloxazine (100 nM) inhibited DHA-mediated neuroprotection. The addition of an A2A receptor antagonist ZM241385 (50 nM), or A3 receptor antagonist VUF5574 (1 μM) was ineffective. Taken together, our results indicated that neuroprotective actions of DHA may depend on P2X, A1, and A2B purinergic receptors activation. Our results reinforce the notion that dietary DHA may act as a local purinergic modulator in order to prevent neurodegenerative diseases.
Figures
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

