Fmoc-Sec(Xan)-OH: synthesis and utility of Fmoc selenocysteine SPPS derivatives with acid-labile sidechain protection
- PMID: 25504629
- PMCID: PMC5538775
- DOI: 10.1002/psc.2723
Fmoc-Sec(Xan)-OH: synthesis and utility of Fmoc selenocysteine SPPS derivatives with acid-labile sidechain protection
Abstract
We report here the synthesis of the first selenocysteine SPPS derivatives which bear TFA-labile sidechain protecting groups. New compounds Fmoc-Sec(Xan)-OH and Fmoc-Sec(Trt)-OH are presented as useful and practical alternatives to the traditional Fmoc-Sec-OH derivatives currently available to the peptide chemist. From a bis Fmoc-protected selenocystine precursor, multiple avenues of diselenide reduction were attempted to determine the most effective method for subsequent attachment of the protecting group electrophiles. Our previously reported one-pot reduction methodology was ultimately chosen as the optimal approach toward the synthesis of these novel building blocks, and both were easily obtained in high yield and purity. Fmoc-Sec(Xan)-OH was discovered to be bench-stable for extended timeframes while the corresponding Fmoc-Sec(Trt)-OH derivative appeared to detritylate slowly when not stored at -20 °C. Both Sec derivatives were incorporated into single- and multiple-Sec-containing test peptides in order to ascertain the peptides' deprotection behavior and final form upon TFA cleavage. Single-Sec-containing test peptides were always isolated as their corresponding diselenide dimers, while dual-Sec-containing peptide sequences were afforded exclusively as their intramolecular diselenides.
Keywords: selenocysteine; sidechain protection; solid phase peptide synthesis; xanthenyl protection.
Copyright © 2014 European Peptide Society and John Wiley & Sons, Ltd.
Figures
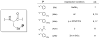
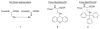
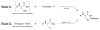


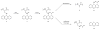
Similar articles
-
The use of 2,2'-dithiobis(5-nitropyridine) (DTNP) for deprotection and diselenide formation in protected selenocysteine-containing peptides.J Pept Sci. 2012 Mar;18(3):155-62. doi: 10.1002/psc.1430. Epub 2012 Jan 16. J Pept Sci. 2012. PMID: 22249911 Free PMC article.
-
Synthetic study on selenocystine-containing peptides.Chem Pharm Bull (Tokyo). 1993 Mar;41(3):502-6. doi: 10.1248/cpb.41.502. Chem Pharm Bull (Tokyo). 1993. PMID: 8477500
-
A comprehensive one-pot synthesis of protected cysteine and selenocysteine SPPS derivatives.Protein Pept Lett. 2014;21(12):1257-64. Protein Pept Lett. 2014. PMID: 24856290
-
Handles for Fmoc solid-phase synthesis of protected peptides.ACS Comb Sci. 2013 May 13;15(5):217-28. doi: 10.1021/co300153c. Epub 2013 Apr 26. ACS Comb Sci. 2013. PMID: 23573835 Review.
-
Advances in Fmoc solid-phase peptide synthesis.J Pept Sci. 2016 Jan;22(1):4-27. doi: 10.1002/psc.2836. J Pept Sci. 2016. PMID: 26785684 Free PMC article. Review.
Cited by
-
Facile removal of 4-methoxybenzyl protecting group from selenocysteine.J Pept Sci. 2019 Oct;25(10):e3209. doi: 10.1002/psc.3209. Epub 2019 Aug 13. J Pept Sci. 2019. PMID: 31410953 Free PMC article.
-
A Simple Zinc-Mediated Method for Selenium Addition to Michael Acceptors.Molecules. 2020 Apr 26;25(9):2018. doi: 10.3390/molecules25092018. Molecules. 2020. PMID: 32357472 Free PMC article.
-
Atom Efficient Preparation of Zinc Selenates for the Synthesis of Selenol Esters under "On Water" Conditions.Molecules. 2017 Jun 8;22(6):953. doi: 10.3390/molecules22060953. Molecules. 2017. PMID: 28594361 Free PMC article.
-
Simple Zn-Mediated Seleno- and Thio-Functionalization of Steroids at C-1 Position.Int J Mol Sci. 2022 Mar 11;23(6):3022. doi: 10.3390/ijms23063022. Int J Mol Sci. 2022. PMID: 35328446 Free PMC article.
-
Water and Aqueous Mixtures as Convenient Alternative Media for Organoselenium Chemistry.Molecules. 2016 Nov 6;21(11):1482. doi: 10.3390/molecules21111482. Molecules. 2016. PMID: 27827970 Free PMC article. Review.
References
-
- Kohrle J. Local activation and inactivation of thyroid hormones: the deiodinase family. Mol Cell Endocrinol. 1999;151:103–19. - PubMed
-
- Lacourciere GM, Levine RL, Stadtman TC. Direct detection of potential selenium delivery proteins by using an Escherichia coli strain unable to incorporate selenium from selenite into proteins. Proceedings of the National Academy of Sciences of the United States of America. 2002;99:9150–3. - PMC - PubMed
-
- Burk RF, Hill KE. Selenoprotein-P - a Selenium-Rich Extracellular Glycoprotein. J Nutr. 1994;124:1891–7. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous
