Methamphetamine causes acute hyperthermia-dependent liver damage
- PMID: 25505562
- PMCID: PMC4184573
- DOI: 10.1002/prp2.8
Methamphetamine causes acute hyperthermia-dependent liver damage
Abstract
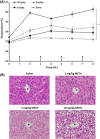
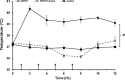
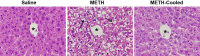
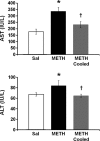
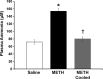
Methamphetamine-induced neurotoxicity has been correlated with damage to the liver but this damage has not been extensively characterized. Moreover, the mechanism by which the drug contributes to liver damage is unknown. This study characterizes the hepatocellular toxicity of methamphetamine and examines if hyperthermia contributes to this liver damage. Livers from methamphetamine-treated rats were examined using electron microscopy and hematoxylin and eosin staining. Methamphetamine increased glycogen stores, mitochondrial aggregation, microvesicular lipid, and hydropic change. These changes were diffuse throughout the hepatic lobule, as evidenced by a lack of hematoxylin and eosin staining. To confirm if these changes were indicative of damage, serum aspartate and alanine aminotransferase were measured. The functional significance of methamphetamine-induced liver damage was also examined by measuring plasma ammonia. To examine the contribution of hyperthermia to this damage, methamphetamine-treated rats were cooled during and after drug treatment by cooling their external environment. Serum aspartate and alanine aminotransferase, as well as plasma ammonia were increased concurrently with these morphologic changes and were prevented when methamphetamine-induced hyperthermia was blocked. These findings support that methamphetamine produces changes in hepatocellular morphology and damage persisting for at least 24 h after drug exposure. At this same time point, methamphetamine treatment significantly increases plasma ammonia concentrations, consistent with impaired ammonia metabolism and functional liver damage. Methamphetamine-induced hyperthermia contributes significantly to the persistent liver damage and increases in peripheral ammonia produced by the drug.
Keywords: Ammonia; hyperthermia; liver damage; methamphetamine.
Figures

Similar articles
-
Peripheral ammonia and blood brain barrier structure and function after methamphetamine.Neuropharmacology. 2016 Aug;107:18-26. doi: 10.1016/j.neuropharm.2016.03.018. Epub 2016 Mar 10. Neuropharmacology. 2016. PMID: 26972828 Free PMC article.
-
Methamphetamine-induced hyperthermia and dopaminergic neurotoxicity in mice: pharmacological profile of protective and nonprotective agents.J Pharmacol Exp Ther. 1995 Dec;275(3):1104-14. J Pharmacol Exp Ther. 1995. PMID: 8531070
-
[Histopathological study on acute poisoning of methamphetamine, morphine or cocaine].Nihon Arukoru Yakubutsu Igakkai Zasshi. 1997 Apr;32(2):122-38. Nihon Arukoru Yakubutsu Igakkai Zasshi. 1997. PMID: 9168637 Japanese.
-
NTP technical report on the toxicity studies of Dibutyl Phthalate (CAS No. 84-74-2) Administered in Feed to F344/N Rats and B6C3F1 Mice.Toxic Rep Ser. 1995 Apr;30:1-G5. Toxic Rep Ser. 1995. PMID: 12209194
-
Splanchnic circulation and metabolism in patients with acute liver failure.Dan Med Bull. 2002 Aug;49(3):177-93. Dan Med Bull. 2002. PMID: 12238280 Review.
Cited by
-
Silencing the Tlr4 Gene Alleviates Methamphetamine-Induced Hepatotoxicity by Inhibiting Lipopolysaccharide-Mediated Inflammation in Mice.Int J Mol Sci. 2022 Jun 18;23(12):6810. doi: 10.3390/ijms23126810. Int J Mol Sci. 2022. PMID: 35743253 Free PMC article.
-
Assessing the role of dopamine in the differential neurotoxicity patterns of methamphetamine, mephedrone, methcathinone and 4-methylmethamphetamine.Neuropharmacology. 2018 May 15;134(Pt A):46-56. doi: 10.1016/j.neuropharm.2017.08.033. Epub 2017 Aug 26. Neuropharmacology. 2018. PMID: 28851615 Free PMC article.
-
Neuronal and peripheral damages induced by synthetic psychoactive substances: an update of recent findings from human and animal studies.Neural Regen Res. 2020 May;15(5):802-816. doi: 10.4103/1673-5374.268895. Neural Regen Res. 2020. PMID: 31719240 Free PMC article. Review.
-
Effect of methamphetamine on the fasting blood glucose in methamphetamine abusers.Metab Brain Dis. 2018 Oct;33(5):1585-1597. doi: 10.1007/s11011-018-0265-8. Epub 2018 Jun 26. Metab Brain Dis. 2018. PMID: 29946958
-
High ambient temperature increases the toxicity and lethality of 3,4-methylenedioxymethamphetamine and methcathinone.Pharmacol Biochem Behav. 2020 May;192:172912. doi: 10.1016/j.pbb.2020.172912. Epub 2020 Mar 19. Pharmacol Biochem Behav. 2020. PMID: 32201298 Free PMC article.
References
-
- Adeva MM, Souto G, Blanco N, Donapetry C. Ammonium metabolism in humans. Metabolism. 2012;61:1495–1511. - PubMed
-
- Ago M, Ago K, Hara K, Kashimura S, Ogata M. Toxicological and histopathological analysis of a patient who died nine days after a single intravenous dose of methamphetamine: a case report. Leg Med (Tokyo) 2006;8:235–239. - PubMed
-
- Albers DS, Sonsalla PK. Methamphetamine-induced hyperthermia and dopaminergic neurotoxicity in mice: pharmacological profile of protective and nonprotective agents. J Pharmacol Exp Ther. 1995;275:1104–1114. - PubMed
-
- Batt AM, Ferrari L. Manifestations of chemically induced liver damage. Clin Chem. 1995;41:1882–1887. - PubMed
-
- Benamar K, Geller EB, Adler MW. A new brain area affected by 3,4-methylenedioxymethamphetamine: a microdialysis-biotelemetry study. Eur J Pharmacol. 2008;596:84–88. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

