Mapping the sevoflurane-binding sites of calmodulin
- PMID: 25505574
- PMCID: PMC4186402
- DOI: 10.1002/prp2.25
Mapping the sevoflurane-binding sites of calmodulin
Abstract
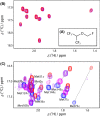
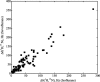
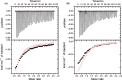
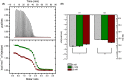
General anesthetics, with sevoflurane (SF) being the first choice inhalational anesthetic agent, provide reversible, broad depressor effects on the nervous system yet have a narrow margin of safety. As characterization of low-affinity binding interactions of volatile substances is exceptionally challenging with the existing methods, none of the numerous cellular targets proposed as chief protagonists in anesthesia could yet be confirmed. The recognition that most critical functions modulated by volatile anesthetics are under the control of intracellular Ca(2+) concentration, which in turn is primarily regulated by calmodulin (CaM), motivated us for characterization of the SF-CaM interaction. Solution NMR (Nuclear Magnetic Resonance) spectroscopy was used to identify SF-binding sites using chemical shift displacement, NOESY and heteronuclear Overhauser enhancement spectroscopy (HOESY) experiments. Binding affinities were measured using ITC (isothermal titration calorimetry). SF binds to both lobes of (Ca(2+))4-CaM with low mmol/L affinity whereas no interaction was observed in the absence of Ca(2+). SF does not affect the calcium binding of CaM. The structurally closely related SF and isoflurane are shown to bind to the same clefts. The SF-binding clefts overlap with the binding sites of physiologically relevant ion channels and bioactive small molecules, but the binding affinity suggests it could only interfere with very weak CaM targets.
Keywords: Anesthesia; ITC; NMR; anesthetic binding; calmodulin; sevoflurane.
Figures

Similar articles
-
Prediction of volatile anesthetic binding sites in proteins.Biophys J. 2006 Nov 1;91(9):3405-14. doi: 10.1529/biophysj.106.082586. Epub 2006 Jul 28. Biophys J. 2006. PMID: 16877516 Free PMC article.
-
Volatile anesthetics inhibit the activity of calmodulin by interacting with its hydrophobic site.Chin Med J (Engl). 2012 Sep;125(17):3166-70. Chin Med J (Engl). 2012. PMID: 22932200
-
Binding of La3+ to calmodulin and its effects on the interaction between calmodulin and calmodulin binding peptide, polistes mastoparan.Biochemistry. 2004 Mar 16;43(10):2688-98. doi: 10.1021/bi035784i. Biochemistry. 2004. PMID: 15005604
-
Al(3+) interaction sites of calmodulin and the Al(3+) effect on target binding of calmodulin.Biochem Biophys Res Commun. 2005 Aug 12;333(4):1060-5. doi: 10.1016/j.bbrc.2005.06.016. Biochem Biophys Res Commun. 2005. PMID: 15964544
-
Structure, dynamics and interaction with kinase targets: computer simulations of calmodulin.Biochim Biophys Acta. 2004 Mar 11;1697(1-2):289-300. doi: 10.1016/j.bbapap.2003.11.032. Biochim Biophys Acta. 2004. PMID: 15023369 Review.
Cited by
-
Calcineurin/P-ERK/Egr-1 Pathway is Involved in Fear Memory Impairment after Isoflurane Exposure in Mice.Sci Rep. 2017 Oct 24;7(1):13947. doi: 10.1038/s41598-017-13975-z. Sci Rep. 2017. PMID: 29066839 Free PMC article.
-
Paramagnetic Ligand Tagging To Identify Protein Binding Sites.J Am Chem Soc. 2015 Sep 9;137(35):11391-8. doi: 10.1021/jacs.5b06220. Epub 2015 Aug 27. J Am Chem Soc. 2015. PMID: 26289584 Free PMC article.
References
-
- Arhem P, Klement G, Nilsson J. Mechanisms of anesthesia: towards integrating network, cellular, and molecular level modeling. Neuropsychopharm. 2003;28:S40–S47. - PubMed
-
- Aulabaugh A, Niemczura WP, Gibbons WA. High-field proton NMR-studies of tryptic fragments of camodulin - a comparison with the native protein. Biochem Biophys Res Commun. 1984;118:225–232. - PubMed
-
- Babu YS, Bugg CE, Cook WJ. Structure of calmodulin refined at 2.2 Å resolution. J Mol Biol. 1988;204:191–204. - PubMed
-
- Bauer W. Pulsed field gradient ‘inverse’ HOESY applied to the isotope pairs 1H,31P and 1H,7Li. Magn Reson Chem. 1996;34:532–537.
-
- Baldisseri DM, Pelton JG, Sparks SW, Torchia DA. Complete 1H and 13C assignment of Lys and Leu sidechains of staphylococcal nuclease using HCCH-COSY and HCCH-TOCSY 3D NMR spectroscopy. FEBS Lett. 1991;281:33–38. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

