A clinical therapeutic protein drug-drug interaction study: coadministration of denosumab and midazolam in postmenopausal women with osteoporosis
- PMID: 25505582
- PMCID: PMC4184705
- DOI: 10.1002/prp2.33
A clinical therapeutic protein drug-drug interaction study: coadministration of denosumab and midazolam in postmenopausal women with osteoporosis
Abstract
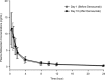
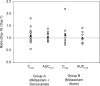
Drug-disease interactions involving therapeutic proteins that target cytokines and potentially impact cytochrome P450 (CYP) enzymes have been of increased interest to drug regulatory agencies and industry sponsors in recent years. This parallel-group open-label study evaluated the effects of the monoclonal antibody denosumab, an inhibitor of the cytokine RANKL, on the pharmacokinetics of the probe CYP3A4 substrate midazolam in postmenopausal women with osteoporosis. The pharmacokinetics of a 2 mg oral dose of midazolam was evaluated on days 1 and 16. Subjects in Group A received a 60 mg subcutaneous dose of denosumab on day 2, 2 weeks before the second midazolam dose, while subjects in Group B did not. For Group A (n = 17), point estimates for the ratio of least square means for midazolam exposures based on maximum observed plasma concentration (C max) and areas under the plasma concentration-time curve (AUCs) on day 16 versus day 1 ranged from 1.02 to 1.04 and 90% confidence intervals were within 0.80-1.25. No period effect was observed for Group B (n = 8). Midazolam and denosumab coadministration was safe and well tolerated. Inhibition of the cytokine RANKL by denosumab does not affect CYP3A4 in postmenopausal women with osteoporosis and will not alter the pharmacokinetics of drugs metabolized by this enzyme. These results are consistent with data suggesting that RANKL does not impact markers of inflammation and represent the first clinical data demonstrating a lack of effect on CYP3A4 of a therapeutic protein that is a cytokine modulator.
Keywords: CYP; cytochrome P450; cytokine modulator; denosumab; drug–drug interaction; menopause; midazolam; postmenopausal; therapeutic protein.
Figures

Similar articles
-
Pharmacokinetic drug interactions between clobazam and drugs metabolized by cytochrome P450 isoenzymes.Pharmacotherapy. 2012 Apr;32(4):340-53. doi: 10.1002/j.1875-9114.2012.01028.x. Epub 2012 Mar 15. Pharmacotherapy. 2012. PMID: 22422635 Clinical Trial.
-
The effect of multiple-dose, oral rifaximin on the pharmacokinetics of intravenous and oral midazolam in healthy volunteers.Pharmacotherapy. 2007 Oct;27(10):1361-9. doi: 10.1592/phco.27.10.1361. Pharmacotherapy. 2007. PMID: 17896891 Clinical Trial.
-
Interaction of sorafenib and cytochrome P450 isoenzymes in patients with advanced melanoma: a phase I/II pharmacokinetic interaction study.Cancer Chemother Pharmacol. 2011 Nov;68(5):1111-8. doi: 10.1007/s00280-011-1585-0. Epub 2011 Feb 25. Cancer Chemother Pharmacol. 2011. PMID: 21350850 Clinical Trial.
-
Role of RANK ligand and denosumab, a targeted RANK ligand inhibitor, in bone health and osteoporosis: a review of preclinical and clinical data.Clin Ther. 2012 Mar;34(3):521-36. doi: 10.1016/j.clinthera.2012.02.002. Clin Ther. 2012. PMID: 22440513 Review.
-
Denosumab--an emerging treatment for postmenopausal osteoporosis.Expert Opin Biol Ther. 2010 Mar;10(3):467-76. doi: 10.1517/14712591003604708. Expert Opin Biol Ther. 2010. PMID: 20095877 Review.
Cited by
-
Effect of Gevokizumab on Interleukin-1β-Mediated Cytochrome P450 3A4 and Drug Transporter Repression in Cultured Human Hepatocytes.Eur J Drug Metab Pharmacokinet. 2017 Oct;42(5):871-878. doi: 10.1007/s13318-017-0406-1. Eur J Drug Metab Pharmacokinet. 2017. PMID: 28260174
-
Assessment of Disease-Related Therapeutic Protein Drug-Drug Interaction for Etrolizumab in Patients With Moderately to Severely Active Ulcerative Colitis.J Clin Pharmacol. 2016 Jun;56(6):693-704. doi: 10.1002/jcph.649. Epub 2016 Jan 11. J Clin Pharmacol. 2016. PMID: 26412221 Free PMC article. Clinical Trial.
-
Effects of Repeated Oral Administration of Esaxerenone on the Pharmacokinetics of Midazolam in Healthy Japanese Males.Eur J Drug Metab Pharmacokinet. 2021 Sep;46(5):685-694. doi: 10.1007/s13318-021-00701-4. Epub 2021 Aug 12. Eur J Drug Metab Pharmacokinet. 2021. PMID: 34383278 Free PMC article. Clinical Trial.
-
Therapeutic protein-drug interaction assessment for daclizumab high-yield process in patients with multiple sclerosis using a cocktail approach.Br J Clin Pharmacol. 2016 Jul;82(1):160-7. doi: 10.1111/bcp.12936. Epub 2016 May 4. Br J Clin Pharmacol. 2016. PMID: 26991517 Free PMC article. Clinical Trial.
-
Evaluation of Potential Disease-Mediated Drug-Drug Interaction in Patients With Moderate-to-Severe Atopic Dermatitis Receiving Dupilumab.Clin Pharmacol Ther. 2018 Dec;104(6):1146-1154. doi: 10.1002/cpt.1058. Epub 2018 Apr 2. Clin Pharmacol Ther. 2018. PMID: 29498038 Free PMC article. Clinical Trial.
References
-
- Abdel-Razzak Z, Loyer P, Fautrel A, Gautier JC, Corcos L, Turlin B, et al. Cytokines down-regulate expression of major cytochrome P-450 enzymes in adult human hepatocytes in primary culture. Mol Pharmacol. 1993;44:707–715. - PubMed
-
- Aitken AE, Richardson TA, Morgan ET. Regulation of drug-metabolizing enzymes and transporters in inflammation. Annu Rev Pharmacol Toxicol. 2006;46:123–149. - PubMed
-
- Anderson DM, Maraskovsky E, Billingsley WL, Dougall WC, Tometsko ME, Roux ER, et al. A homologue of the TNF receptor and its ligand enhance T-cell growth and dendritic-cell function. Nature. 1997;390:175–179. - PubMed
-
- Bekker PJ, Holloway DL, Rasmussen AS, Murphy R, Martin SW, Leese PT, et al. A single-dose placebo-controlled study of AMG 162, a fully human monoclonal antibody to RANKL, in postmenopausal women. J Bone Miner Res. 2004;19:1059–1066. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

