Assessing the heterogeneity of treatment effects via potential outcomes of individual patients
- PMID: 25506088
- PMCID: PMC4261197
- DOI: 10.1111/rssc.12012
Assessing the heterogeneity of treatment effects via potential outcomes of individual patients
Abstract
There is growing interest in understanding the heterogeneity of treatment effects (HTE), which has important implications in treatment evaluation and selection. The standard approach to assessing HTE (i.e. subgroup analyses based on known effect modifiers) is informative about the heterogeneity between subpopulations but not within. It is arguably more informative to assess HTE in terms of individual treatment effects, which can be defined by using potential outcomes. However, estimation of HTE based on potential outcomes is challenged by the lack of complete identifiability. The paper proposes methods to deal with the identifiability problem by using relevant information in baseline covariates and repeated measurements. If a set of covariates is sufficient for explaining the dependence between potential outcomes, the joint distribution of potential outcomes and hence all measures of HTE will then be identified under a conditional independence assumption. Possible violations of this assumption can be addressed by including a random effect to account for residual dependence or by specifying the conditional dependence structure directly. The methods proposed are shown to reduce effectively the uncertainty about HTE in a trial of human immunodeficiency virus.
Keywords: Causal inference; Conditional independence; Copula; Counterfactual; Random effect; Sensitivity analysis.
Figures
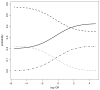
 , π00;— — —, π01; · · · · · ·, π10; · — · —, π11
, π00;— — —, π01; · · · · · ·, π10; · — · —, π11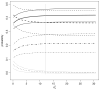
 , — — —, · — · —, · · · · · · ·) and 95% confidence intervals (
, — — —, · — · —, · · · · · · ·) and 95% confidence intervals (
 , — — —, · — · —, · · · · · · ·) for π00 (
, — — —, · — · —, · · · · · · ·) for π00 (
 ),π01(— — —),π10(· · · · · · ·) and π11 (· — · —)
),π01(— — —),π10(· · · · · · ·) and π11 (· — · —)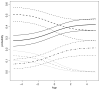
 , — — —, · — · —, · · · · · · ·) and 95% confidence intervals (
, — — —, · — · —, · · · · · · ·) and 95% confidence intervals (
 , — — —, ·— · —, ·· · · · ·) for π00 (
, — — —, ·— · —, ·· · · · ·) for π00 (
 ), π01 (— — —), π10 (· · · · · · ·) and π11 (· — · —), as functions of ρ(X) ≡ ρ
), π01 (— — —), π10 (· · · · · · ·) and π11 (· — · —), as functions of ρ(X) ≡ ρSimilar articles
-
Developing Software for Using Bayesian Regression to Evaluate Heterogeneity of Treatment Effects in Data from Randomized Controlled Trials [Internet].Washington (DC): Patient-Centered Outcomes Research Institute (PCORI); 2018 Jul. Washington (DC): Patient-Centered Outcomes Research Institute (PCORI); 2018 Jul. PMID: 38620392 Free Books & Documents. Review.
-
Assessing Heterogeneity of Treatment Effect in Real-World Data.Ann Intern Med. 2023 Apr;176(4):536-544. doi: 10.7326/M22-1510. Epub 2023 Mar 21. Ann Intern Med. 2023. PMID: 36940440 Free PMC article.
-
Characterizing Treatment Effect Heterogeneity Using Real-World Data.Clin Pharmacol Ther. 2025 May;117(5):1209-1216. doi: 10.1002/cpt.3627. Epub 2025 Mar 6. Clin Pharmacol Ther. 2025. PMID: 40047339 Free PMC article. Review.
-
Preventing false discovery of heterogeneous treatment effect subgroups in randomized trials.Trials. 2018 Jul 16;19(1):382. doi: 10.1186/s13063-018-2774-5. Trials. 2018. PMID: 30012181 Free PMC article.
-
The Use of Covariates and Random Effects in Evaluating Predictive Biomarkers Under a Potential Outcome Framework.Ann Appl Stat. 2014 Dec;8(4):2336-2355. doi: 10.1214/14-AOAS773. Epub 2014 Dec 19. Ann Appl Stat. 2014. PMID: 26779295 Free PMC article.
Cited by
-
Joint mixed-effects models for causal inference with longitudinal data.Stat Med. 2018 Feb 28;37(5):829-846. doi: 10.1002/sim.7567. Epub 2017 Dec 4. Stat Med. 2018. PMID: 29205454 Free PMC article.
-
Capturing heterogeneity in repeated measures data by fusion penalty.Stat Med. 2021 Apr 15;40(8):1901-1916. doi: 10.1002/sim.8878. Epub 2021 Jan 31. Stat Med. 2021. PMID: 33517583 Free PMC article.
-
Inequality in treatment benefits: Can we determine if a new treatment benefits the many or the few?Biostatistics. 2017 Apr 1;18(2):308-324. doi: 10.1093/biostatistics/kxw049. Biostatistics. 2017. PMID: 28025183 Free PMC article.
-
Characterizing expected benefits of biomarkers in treatment selection.Biostatistics. 2015 Apr;16(2):383-99. doi: 10.1093/biostatistics/kxu039. Epub 2014 Sep 3. Biostatistics. 2015. PMID: 25190512 Free PMC article.
-
Adaptive Enrichment Designs in Clinical Trials.Annu Rev Stat Appl. 2021 Mar;8(1):393-411. doi: 10.1146/annurev-statistics-040720-032818. Annu Rev Stat Appl. 2021. PMID: 36212769 Free PMC article.
References
-
- Berger RL, Boos DD. P values maximised over a confidence set for the nuisance parameter. J Am Statist Ass. 1994;89:1012–1016.
-
- Carroll RJ, Spiegelman CH, Lan KKG, Kent KT, Abbott RD. On error-in-variables for binary regression models. Biometrika. 1984;71:19–25.
-
- Fatkenheuer G, Nelson M, Lazzarin A, Konourina I, Hoepelman AI, Lampiris H, Hirschel B, Tebas P, Raffi F, Trottier B, Bellos N, Saag M, Cooper DA, Westby M, Tawadrous M, Sullivan JF, Ridgway C, Dunne MW, Felstead S, Mayer H, van der Ryst E, MOTIVATE Study Teams Subgroup analyses of maraviroc in previously treated R5 HIV-1 infection. New Engl J Med. 2008;359:1442–1455. - PubMed
-
- Gadbury GL, Iyer HK. Unit-treatment interaction and its practical consequences. Biometrics. 2000;56:882–885. - PubMed
-
- Gadbury GL, Iyer HK, Albert JM. Individual treatment effects in randomized trials with binary outcomes. J Statist Planng Inf. 2004;121:163–174.
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
