Interaction between endoplasmic/sarcoplasmic reticulum stress (ER/SR stress), mitochondrial signaling and Ca(2+) regulation in airway smooth muscle (ASM)
- PMID: 25506723
- PMCID: PMC4386727
- DOI: 10.1139/cjpp-2014-0361
Interaction between endoplasmic/sarcoplasmic reticulum stress (ER/SR stress), mitochondrial signaling and Ca(2+) regulation in airway smooth muscle (ASM)
Abstract
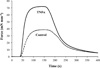
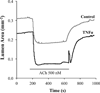
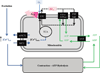
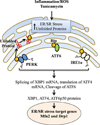
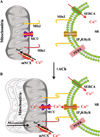
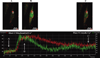
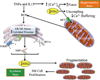
Airway inflammation is a key aspect of diseases such as asthma. Several inflammatory cytokines (e.g., TNFα and IL-13) increase cytosolic Ca(2+) ([Ca(2+)]cyt) responses to agonist stimulation and Ca(2+) sensitivity of force generation, thereby enhancing airway smooth muscle (ASM) contractility (hyper-reactive state). Inflammation also induces ASM proliferation and remodeling (synthetic state). In normal ASM, the transient elevation of [Ca(2+)]cyt induced by agonists leads to a transient increase in mitochondrial Ca(2+) ([Ca(2+)]mito) that may be important in matching ATP production with ATP consumption. In human ASM (hASM) exposed to TNFα and IL-13, the transient increase in [Ca(2+)]mito is blunted despite enhanced [Ca(2+)]cyt responses. We also found that TNFα and IL-13 induce reactive oxidant species (ROS) formation and endoplasmic/sarcoplasmic reticulum (ER/SR) stress (unfolded protein response) in hASM. ER/SR stress in hASM is associated with disruption of mitochondrial coupling with the ER/SR membrane, which relates to reduced mitofusin 2 (Mfn2) expression. Thus, in hASM it appears that TNFα and IL-13 result in ROS formation leading to ER/SR stress, reduced Mfn2 expression, disruption of mitochondrion-ER/SR coupling, decreased mitochondrial Ca(2+) buffering, mitochondrial fragmentation, and increased cell proliferation.
Keywords: ER/SR stress; airway; asthma; asthme; inflammation; réponse aux protéines dépliées; stress du RE/RS; unfolded protein response; voies respiratoires.
Figures



Similar articles
-
Inflammation-Induced Protein Unfolding in Airway Smooth Muscle Triggers a Homeostatic Response in Mitochondria.Int J Mol Sci. 2020 Dec 31;22(1):363. doi: 10.3390/ijms22010363. Int J Mol Sci. 2020. PMID: 33396378 Free PMC article. Review.
-
TNFα decreases mitochondrial movement in human airway smooth muscle.Am J Physiol Lung Cell Mol Physiol. 2017 Jul 1;313(1):L166-L176. doi: 10.1152/ajplung.00538.2016. Epub 2017 May 4. Am J Physiol Lung Cell Mol Physiol. 2017. PMID: 28473328 Free PMC article.
-
TNFα selectively activates the IRE1α/XBP1 endoplasmic reticulum stress pathway in human airway smooth muscle cells.Am J Physiol Lung Cell Mol Physiol. 2020 Mar 1;318(3):L483-L493. doi: 10.1152/ajplung.00212.2019. Epub 2020 Jan 15. Am J Physiol Lung Cell Mol Physiol. 2020. PMID: 31940218 Free PMC article.
-
Inflammation alters regional mitochondrial Ca²+ in human airway smooth muscle cells.Am J Physiol Cell Physiol. 2012 Aug 1;303(3):C244-56. doi: 10.1152/ajpcell.00414.2011. Epub 2012 Jun 6. Am J Physiol Cell Physiol. 2012. PMID: 22673614 Free PMC article.
-
Microdomain elements of airway smooth muscle in calcium regulation and cell proliferation.J Physiol Pharmacol. 2018 Apr;69(2). doi: 10.26402/jpp.2018.2.01. Epub 2018 Jun 13. J Physiol Pharmacol. 2018. PMID: 29920471 Review.
Cited by
-
Emerging concepts in smooth muscle contributions to airway structure and function: implications for health and disease.Am J Physiol Lung Cell Mol Physiol. 2016 Dec 1;311(6):L1113-L1140. doi: 10.1152/ajplung.00370.2016. Epub 2016 Oct 14. Am J Physiol Lung Cell Mol Physiol. 2016. PMID: 27742732 Free PMC article. Review.
-
Mitochondrial regulation of airway smooth muscle functions in health and pulmonary diseases.Arch Biochem Biophys. 2019 Mar 15;663:109-119. doi: 10.1016/j.abb.2019.01.002. Epub 2019 Jan 8. Arch Biochem Biophys. 2019. PMID: 30629957 Free PMC article. Review.
-
Update on the role of endoplasmic reticulum stress in asthma.Am J Transl Res. 2020 Apr 15;12(4):1168-1183. eCollection 2020. Am J Transl Res. 2020. PMID: 32355534 Free PMC article. Review.
-
STIM1 is a core trigger of airway smooth muscle remodeling and hyperresponsiveness in asthma.Proc Natl Acad Sci U S A. 2022 Jan 4;119(1):e2114557118. doi: 10.1073/pnas.2114557118. Proc Natl Acad Sci U S A. 2022. PMID: 34949717 Free PMC article.
-
Bronchial smooth muscle cell in asthma: where does it fit?BMJ Open Respir Res. 2022 Sep;9(1):e001351. doi: 10.1136/bmjresp-2022-001351. BMJ Open Respir Res. 2022. PMID: 36109087 Free PMC article. Review.
References
-
- Amrani Y. Modulation of airway smooth muscle contractile function by TNFα and IL-13 and airway hyperresponsiveness in asthma. In: Wang Y-X, editor. Calcium signaling in airway smooth muscle cells. Springer International Publishing; 2014. pp. 423–439.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

