A zebrafish model of myelodysplastic syndrome produced through tet2 genomic editing
- PMID: 25512612
- PMCID: PMC4323485
- DOI: 10.1128/MCB.00971-14
A zebrafish model of myelodysplastic syndrome produced through tet2 genomic editing
Abstract
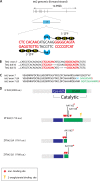
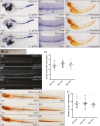
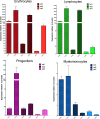
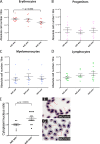
The ten-eleven translocation 2 gene (TET2) encodes a member of the TET family of DNA methylcytosine oxidases that converts 5-methylcytosine (5mC) to 5-hydroxymethylcytosine (5hmC) to initiate the demethylation of DNA within genomic CpG islands. Somatic loss-of-function mutations of TET2 are frequently observed in human myelodysplastic syndrome (MDS), which is a clonal malignancy characterized by dysplastic changes of developing blood cell progenitors, leading to ineffective hematopoiesis. We used genome-editing technology to disrupt the zebrafish Tet2 catalytic domain. tet2(m/m) (homozygous for the mutation) zebrafish exhibited normal embryonic and larval hematopoiesis but developed progressive clonal myelodysplasia as they aged, culminating in myelodysplastic syndromes (MDS) at 24 months of age, with dysplasia of myeloid progenitor cells and anemia with abnormal circulating erythrocytes. The resultant tet2(m/m) mutant zebrafish lines show decreased levels of 5hmC in hematopoietic cells of the kidney marrow but not in other cell types, most likely reflecting the ability of other Tet family members to provide this enzymatic activity in nonhematopoietic tissues but not in hematopoietic cells. tet2(m/m) zebrafish are viable and fertile, providing an ideal model to dissect altered pathways in hematopoietic cells and, for small-molecule screens in embryos, to identify compounds with specific activity against tet2 mutant cells.
Copyright © 2015, American Society for Microbiology. All Rights Reserved.
Figures






References
-
- Tefferi A, Lim KH, Abdel-Wahab O, Lasho TL, Patel J, Patnaik MM, Hanson CA, Pardanani A, Gilliland DG, Levine RL. 2009. Detection of mutant TET2 in myeloid malignancies other than myeloproliferative neoplasms: CMML, MDS, MDS/MPN and AML. Leukemia 23:1343–1345. doi:10.1038/leu.2009.59. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous
