The Na⁺/H⁺ exchanger (NHE1) as a novel co-adjuvant target in paclitaxel therapy of triple-negative breast cancer cells
- PMID: 25514463
- PMCID: PMC4359231
- DOI: 10.18632/oncotarget.2860
The Na⁺/H⁺ exchanger (NHE1) as a novel co-adjuvant target in paclitaxel therapy of triple-negative breast cancer cells
Abstract
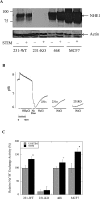
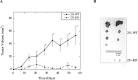
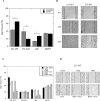
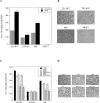
Dysregulation of Na⁺/H⁺ exchanger isoform one (NHE1) activity is a hallmark of cells undergoing tumorigenesis and metastasis, the leading cause of patient mortality. The acidic tumor microenvironment is thought to facilitate the development of resistance to chemotherapy drugs and to promote extracellular matrix remodeling leading to metastasis. Here, we investigated NHE1 as a co-adjuvant target in paclitaxel chemotherapy of metastatic breast cancer. We generated a stable NHE1-knockout of the highly invasive, triple-negative, MDA-MB-231 breast cancer cells. The NHE1-knockout cells proliferated comparably to parental cells, but had markedly lower rates of migration and invasion in vitro. In vivo xenograft tumor growth in athymic nude mice was also dramatically decreased compared to parental MDA-MB-231 cells. Loss of NHE1 expression also increased the susceptibility of knockout cells to paclitaxel-mediated cell death. NHE1 inhibition, in combination with paclitaxel, resulted in a dramatic decrease in viability, and migratory and invasive potential of triple-negative breast cancer cells, but not in hormone receptor-positive, luminal MCF7 cells. Our data suggest that NHE1 is critical in triple-negative breast cancer metastasis, and its chemical inhibition boosts the efficacy of paclitaxel in vitro, highlighting NHE1 as a novel, potential co-adjuvant target in breast cancer chemotherapy.
Figures



References
-
- Fliegel L. The Na+ /H+ exchanger isoform 1. Int J Biochem Cell Biol. 2005;37(1):33–37. - PubMed
-
- Bkaily G, Nader M, Avedanian L, Jacques D, Perrault C, Abdel-Samad D, D'Orleans-Juste P, Gobeil F, Hazzouri KM. Immunofluorescence revealed the presence of NHE-1 in the nuclear membranes of rat cardiomyocytes and isolated nuclei of human, rabbit, and rat aortic and liver tissues. Can J Physiol Pharmacol. 2004;82(8-9):805–811. - PubMed
-
- Malo ME, Fliegel L. Physiological role and regulation of the Na+ /H+ exchanger. Can J Physiol Pharmacol. 2006;84(11):1081–1095. - PubMed
-
- Amith SR, Fliegel L. Regulation of the Na+ /H+ Exchanger (NHE1) in Breast Cancer Metastasis. Cancer research. 2013;73(4):1259–1264. - PubMed
-
- Webb BA, Chimenti M, Jacobson MP, Barber DL. Dysregulated pH: a perfect storm for cancer progression. Nat Rev Cancer. 2011;11(9):671–677. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

