Premyelinated central axons express neurotoxic NMDA receptors: relevance to early developing white-matter injury
- PMID: 25515212
- PMCID: PMC4420873
- DOI: 10.1038/jcbfm.2014.227
Premyelinated central axons express neurotoxic NMDA receptors: relevance to early developing white-matter injury
Abstract
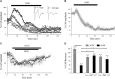
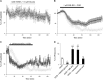
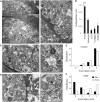
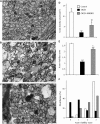
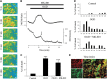
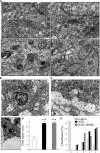
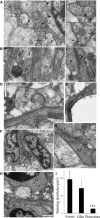
Ischemic-type injury to developing white matter is associated with the significant clinical condition cerebral palsy and with the cognitive deficits associated with premature birth. Premyelinated axons are the major cellular component of fetal white matter and loss of axon function underlies the disability, but the cellular mechanisms producing ischemic injury to premyelinated axons have not previously been described. Injury was found to require longer periods of modelled ischemia than at latter developmental points. Ischemia produced initial hyperexcitability in axons followed by loss of function after Na(+) and Ca(2+) influx. N-methyl-D-aspartate- (NMDA) type glutamate receptor (GluR) agonists potentiated axon injury while antagonists were protective. The NMDA GluR obligatory Nr1 subunit colocalized with markers of small premyelinated axons and expression was found at focal regions of axon injury. Ischemic injury of glial cells present in early developing white matter was NMDA GluR independent. Axons in human postconception week 18 to 23 white matter had a uniform prediameter expansion phenotype and postembedded immuno-gold labelling showed Nr1 subunit expression on the membrane of these axons, demonstrating a shared key neuropathologic feature with the rodent model. Premyelinated central axons therefore express high levels of functional NMDA GluRs that confer sensitivity to ischemic injury.
Figures
References
-
- Herkert M, Rottger S, Becker CM. The NMDA receptor subunit NR2B of neonatal rat brain: complex formation and enrichment in axonal growth cones. Eur J Neurosci. 1998;10:1553–1562. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

