SPRY1 promotes the degradation of uPAR and inhibits uPAR-mediated cell adhesion and proliferation
- PMID: 25520860
- PMCID: PMC4266704
SPRY1 promotes the degradation of uPAR and inhibits uPAR-mediated cell adhesion and proliferation
Abstract
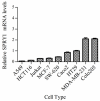
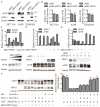
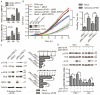
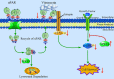
Urokinase plasminogen activator receptor (uPAR) is a GPI anchored cell surface protein that is closely associated with invasion, migration, and metastasis of cancer cells. Many functional extracellular proteins and transmembrane receptors interact with uPAR. However, few studies have examined the association of uPAR with cytoplasm proteins. We previously used yeast two-hybrid screening to isolate several novel uPAR-interacting cytoplasmic proteins, including Sprouty1 (SPRY1), an inhibitor of the (Ras-mitogen-activated protein kinase) MAPK pathway. In this study, we show that SPRY1 interacts with uPAR and directs it toward lysosomal-mediated degradation. Overexpression of SPRY1 decreased the cell surface and cytoplasmic uPAR protein level. Moreover, SPRY1 overexpression augmented uPAR-induced cell adhesion to vitronectin as well as proliferation of cancer cells. Our results also further support the critical role of SPRY1 contribution to tumor growth. In a subcutaneous tumor model, overexpression of SPRY1 in HCT116 or A549 xenograft in athymic nude mice led to great suppression of tumor growth. These results show that SPRY1 may affect tumor cell function through direct interaction with uPAR and promote its lysosomal degradation.
Keywords: SPRY1; adhesion; degradation; proliferation; uPAR.
Figures



References
-
- Alpízar-Alpízar W, Nielsen BS, Sierra R, Illemann M, Ramírez JA, Arias A, Durán S, Skarstein A, Ovrebo K, Lund LR, Laerum OD. Urokinase plasminogen activator receptor is expressed in invasive cells in gastric carcinomas from high- and low-risk countries. Int J Cancer. 2010;126:405–15. - PubMed
-
- Wei Y, Waltz DA, Rao N, Drummond RJ, Rosenberg S, Chapman HA. Identification of the urokinase receptor as an adhesion receptor for vitronectin. J Biol Chem. 1994;269:32380–8. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
