A novel immunocompetent murine model for replicating oncolytic adenoviral therapy
- PMID: 25525035
- PMCID: PMC4298785
- DOI: 10.1038/cgt.2014.64
A novel immunocompetent murine model for replicating oncolytic adenoviral therapy
Abstract
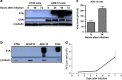
Oncolytic adenoviruses are under investigation as a promising novel strategy for cancer immunotherapeutics. Unfortunately, there is no immunocompetent mouse cancer model to test oncolytic adenovirus because murine cancer cells are generally unable to produce infectious viral progeny from human adenoviruses. We find that the murine K-ras-induced lung adenocarcinoma cell line ADS-12 supports adenoviral infection and generates infectious viral progeny. ADS-12 cells express the coxsackie and adenovirus receptor and infected ADS-12 cells express the viral protein E1A. We find that our previously described oncolytic virus, adenovirus TAV-255 (AdTAV-255), kills ADS-12 cells in a dose- and time-dependent manner. We investigated ADS-12 cells as an in-vivo model system for replicating oncolytic adenoviruses. Subcutaneous injection of ADS-12 cells into immunocompetent 129 mice led to tumor formation in all injected mice. Intratumoral injection of AdTAV-255 in established tumors causes a significant reduction in tumor growth. This model system represents the first fully immunocompetent mouse model for cancer treatment with replicating oncolytic adenoviruses, and therefore will be useful to study the therapeutic effect of oncolytic adenoviruses in general and particularly immunostimulatory viruses designed to evoke an antitumor immune response.
Figures




References
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources

