A THEMIS:SHP1 complex promotes T-cell survival
- PMID: 25535246
- PMCID: PMC4339124
- DOI: 10.15252/embj.201387725
A THEMIS:SHP1 complex promotes T-cell survival
Abstract
THEMIS is critical for conventional T-cell development, but its precise molecular function remains elusive. Here, we show that THEMIS constitutively associates with the phosphatases SHP1 and SHP2. This complex requires the adapter GRB2, which bridges SHP to THEMIS in a Tyr-phosphorylation-independent fashion. Rather, SHP1 and THEMIS engage with the N-SH3 and C-SH3 domains of GRB2, respectively, a configuration that allows GRB2-SH2 to recruit the complex onto LAT. Consistent with THEMIS-mediated recruitment of SHP to the TCR signalosome, THEMIS knock-down increased TCR-induced CD3-ζ phosphorylation, Erk activation and CD69 expression, but not LCK phosphorylation. This generalized TCR signalling increase led to augmented apoptosis, a phenotype mirrored by SHP1 knock-down. Remarkably, a KI mutation of LCK Ser59, previously suggested to be key in ERK-mediated resistance towards SHP1 negative feedback, did not affect TCR signalling nor ligand discrimination in vivo. Thus, the THEMIS:SHP complex dampens early TCR signalling by a previously unknown molecular mechanism that favours T-cell survival. We discuss possible implications of this mechanism in modulating TCR output signals towards conventional T-cell development and differentiation.
Keywords: SHP1; TCR; THEMIS; apoptosis; negative feedback.
© 2014 The Authors. Published under the terms of the CC BY 4.0 license.
Figures

SHP1 immunoprecipitation (IP) from Jurkat 1G4-CD8 cells. Upper panel of immunoblots shows expression levels in the input lysates, and isolated protein complexes are shown at the bottom. Rabbit IgG IP (isotype) is shown as control.
Lentiviral knock-down/re-expression constructs for THEMIS-wt-Strep and THEMIS-dPRR1-Strep in 1G4-CD8 cells. Knock-down/re-expression constructs are based on shTHEMIS-128476. Red arrowheads indicate the specific signal for endogenous THEMIS and re-expressed THEMIS-Strep. The higher molecular weight band observed in all lanes is a non-specific signal.
Streptactin pull-downs (PDs) of THEMIS-Strep from 1G4-CD8 cells. Cells as described in (B) were stimulated with 6V tetramers, followed by Streptactin PDs of THEMIS and immunoblot analysis of isolated protein complexes. Separate experiments for SHP1 and SHP2 are shown.
SHP1 IP in the presence and absence of SHP1 tail phosphorylation. SHP1 was immunoprecipitated from J.CaM1.6 LCK-Tet cells. Where indicated, LCK expression was induced with doxycycline prior to the experiment or Src-kinase inhibitor PP2 was used to abolish residual SHP1-pY564. The upper panel of immunoblots shows the effect of LCK activity on SHP1-pY564 levels. The red arrowhead indicates residual pY564. Isolated protein complexes are shown in the lower panels.
In vitro phosphatase treatment of the THEMIS:GRB2:SHP1 complex. Bead-bound THEMIS-Strep complexes from 1G4-CD8 cells were incubated in alkaline phosphatase (AP) buffer in the absence (lane 1) or presence of AP (lane 2), or left completely untreated (lane 3), prior to washing and elution.
Anti-HA IP from HEK293 cells transfected with HA-SHP1 and GRB2-Myc constructs. GRB2 mutants used: W36K, N-SH3 mutant; W193K, C-SH3 mutant. Upper panels show expression levels in the input; isolated proteins complexes are shown at the bottom. Relative amounts of GRB2-Myc are normalized to the bait HA-SHP1.
Far-Western blot of GRB2-SH3 domains binding to full-length SHP1. HA IPs from empty vector or HA-SHP1-transfected HEK293 cells were subjected to far-Western blotting using recombinant GST-tagged N -or C-SH3 domains of GRB2. GST alone served as a background control. Blots were re-probed for SHP1 loading for normalization of anti-GST signals. Data shown are representative of three independent experiments.

Lentiviral knock-down (KD) of THEMIS expression in 1G4-CD8 cells. The efficiency of the THEMIS KD was assessed using immunoblotting.
Phospho-flow analysis of pERK responses in THEMIS KD cells. 1G4-CD8 control and THEMIS KD cells were stimulated with NY-ESO-1 pMHC tetramers and analysed for ERK phosphorylation using flow cytometry. Data shown are representative of three independent experiments.
Quantification of pERK-positive cells from (B). Data from three independent experiments were used in the analysis. n = 3, means ± SEM are shown; two-tailed unpaired Student's t-test, *P < 0.05, **P < 0.01.
Phospho-flow analysis of pERK responses against titrated concentrations of 9V tetramer. Time point was fixed at 60 s. Data shown are representative of three independent experiments.
Quantification of pERK-positive cells from (D). Data from three independent experiments were used in the analysis. n = 3, means ± SEM are shown.
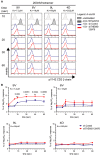
1G4-CD8 control and THEMIS KD cells were stimulated with NY-ESO-1 pMHC tetramers and analysed for pY142-CD3-ζ phosphorylation by flow cytometry. Data shown are representative of three independent experiments.
Quantification of pY142-CD3-ζ responses of cells from (A). Data from three independent experiments were used in the analysis. Responses are expressed as percentage of the positive control sodium pervanadate. n = 3, means ± SEM are shown; two-tailed unpaired Student's t-test, **P < 0.01.
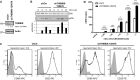
Lentiviral knock-down of THEMIS expression in primary human CD4+ T cells. Following transduction, cells were expanded on CD3/CD28 beads in the presence of exogenous IL-2 and analysed for THEMIS KD by immunoblotting on day 4 post-transduction. Data shown are representative of three independent experiments.
pERK response in THEMIS KD human CD4+ T cells. Transduced CD4+ cells were removed from beads, rested overnight and stimulated with CD3 mAb for the indicated time points. ERK phosphorylation was assessed by immunoblotting. Data shown are representative of three independent experiments.
Expression of CD25 and CD69 surface markers of THEMIS KD human CD4+ T cells was assessed on day 4 post-transduction by flow cytometry. Data shown are representative of three independent experiments.
CD69 up-regulation on re-stimulated THEMIS KD CD4+ T cells. CD3/CD28 beads were removed on day 1 post-transduction, and cells were cultured in IL-2 and IL-7. After removal of cell debris at day 4, cells were restimulated with CD3/CD28 beads at the indicated bead to cell ratio. CD69 surface expression was assessed by flow cytometry 24 h later. Data shown are representative of two independent experiments. n = 3, means ± SD are shown; two-tailed unpaired Student's t-test, *P < 0.05, ***P < 0.001.
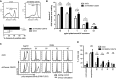
Donor-derived human CD4+ T cells transduced as in Fig4A were analysed for apoptosis by Annexin-V surface staining on day 4 post-transduction. Data from three independent experiments were used in the analysis. n = 3, means ± SD are shown; two-tailed unpaired Student's t-test, **P < 0.01.
Annexin-V staining of THEMIS KD 1G4-CD8 cells. THEMIS KD and control 1G4-CD8 cells were stimulated for 24 h with plate-bound 6V tetramers. Annexin-V surface expression was analysed by flow cytometry and is shown as fold-increase over non-stimulated. Data from three independent experiments were used in the analysis. n = 3, means ± SD are shown; two-tailed unpaired Student's t-test, *P < 0.05, **P < 0.01, ***P < 0.001; ns, not significant.
Poly-caspase activity in THEMIS KD 1G4-CD8 cells. Cells were stimulated for 24 h with NY-ESO-1 pMHC tetramers. Camptothecin was used as a positive control for caspase activation. A FAM-FLICA detection probe was used to assess poly-caspase activity by flow cytometry. Data shown are a representative example of three independent experiments.
Quantification of poly-caspase activity of cells from (C). Data from three independent experiments were used in the analysis. n = 3, means ± SEM are shown; two-tailed unpaired Student's t-test, *P < 0.05, **P < 0.01; ns, not significant.

Lentiviral KD of SHP1 expression in 1G4-CD8 cells. KD efficiency was assessed using immunoblotting.
Annexin-V staining of THEMIS and SHP1 KD 1G4-CD8 cells. Control, THEMIS and SHP1 knock-down Jurkat 1G4-CD8 cells were stimulated for 24 h with plate-bound 6V tetramers. Annexin-V surface expression was analysed by flow cytometry and is shown as fold-increase over non-stimulated. Data from three independent experiments were used in the analysis. n = 3, means ± SD are shown; two-way ANOVA, Bonferroni post-test, *P < 0.05, **P < 0.01, ***P < 0.001.
Lentiviral knock-down of THEMIS and SHP1 expression in primary human CD4+ T cells. Following transduction, cells were expanded on CD3/CD28 beads in the presence of exogenous IL-2. Knock-down efficiency was analysed by immunoblotting on day 4 post-transduction.
Expression of CD25, CD69 and Annexin-V surface markers of cells from (C) was assessed by flow cytometry. Data shown are representative of three independent experiments.
Annexin-V staining of 1G4-CD8 cells re-expressing a SHP1 tail tyrosine mutant. Jurkat 1G4-CD8 cells were transduced with Knock-down/re-expression constructs for HA-SHP1 wild-type (wt) or Tyr536/564 to Phe double mutant (YYFF). Cells were stimulated and analysed by flow cytometry as in (B). Data shown are representative of three independent experiments. n = 3, means ± SD are shown.
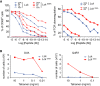
CD8+ T cells from OT-I Rag2−/− mice expressing wild-type (LCK) or LCKS59A proteins were stimulated with H-2Kb-positive antigen-presenting cells pulsed with serial dilutions of the OVA (257–264) peptide, the OVA peptide variants Q4 and T4, and VSV, a peptide that is not recognized by the OT-I TCR. CD69 up-regulation (left) and TCR down-regulation (right) were analysed. Data are representative of three independent experiments.
CD8+ T cells from OT-I Rag2−/− mice expressing wild-type (LCK) or LCKS59A proteins were labelled with CFSE and stimulated with serial dilution of H-2Kb-OVA or H-2Kb-Q4R7 peptide-MHC tetramers. After 3 days, the absolute numbers of divided T cells were determined. Data are representative of three independent experiments.
Similar articles
-
Themis-associated phosphatase activity controls signaling in T cell development.Proc Natl Acad Sci U S A. 2018 Nov 27;115(48):E11331-E11340. doi: 10.1073/pnas.1720209115. Epub 2018 Nov 9. Proc Natl Acad Sci U S A. 2018. PMID: 30413615 Free PMC article.
-
GRB2 promotes thymocyte positive selection by facilitating THEMIS-mediated inactivation of SHP1.J Exp Med. 2023 Jul 3;220(7):e20221649. doi: 10.1084/jem.20221649. Epub 2023 Apr 17. J Exp Med. 2023. PMID: 37067793 Free PMC article.
-
T cell receptor (TCR)-induced tyrosine phosphorylation dynamics identifies THEMIS as a new TCR signalosome component.J Biol Chem. 2011 Mar 4;286(9):7535-47. doi: 10.1074/jbc.M110.201236. Epub 2010 Dec 28. J Biol Chem. 2011. PMID: 21189249 Free PMC article.
-
THEMIS: a critical TCR signal regulator for ligand discrimination.Curr Opin Immunol. 2015 Apr;33:86-92. doi: 10.1016/j.coi.2015.01.020. Epub 2015 Feb 18. Curr Opin Immunol. 2015. PMID: 25700024 Review.
-
TAOK3, a Regulator of LCK-SHP-1 Crosstalk during TCR Signaling.Crit Rev Immunol. 2019;39(1):59-81. doi: 10.1615/CritRevImmunol.2019030480. Crit Rev Immunol. 2019. PMID: 31679194 Review.
Cited by
-
T Cell Costimulation by CD6 Is Dependent on Bivalent Binding of a GADS/SLP-76 Complex.Mol Cell Biol. 2017 May 16;37(11):e00071-17. doi: 10.1128/MCB.00071-17. Print 2017 Jun 1. Mol Cell Biol. 2017. PMID: 28289074 Free PMC article.
-
THEMIS increases TCR signaling in CD4+CD8+ thymocytes by inhibiting the activity of the tyrosine phosphatase SHP1.Sci Signal. 2023 May 9;16(784):eade1274. doi: 10.1126/scisignal.ade1274. Epub 2023 May 9. Sci Signal. 2023. PMID: 37159521 Free PMC article.
-
Adapting T Cell Receptor Ligand Discrimination Capability via LAT.Front Immunol. 2021 Apr 16;12:673196. doi: 10.3389/fimmu.2021.673196. eCollection 2021. Front Immunol. 2021. PMID: 33936119 Free PMC article. Review.
-
The Analysis of PTPN6 for Bladder Cancer: An Exploratory Study Based on TCGA.Dis Markers. 2020 May 13;2020:4312629. doi: 10.1155/2020/4312629. eCollection 2020. Dis Markers. 2020. PMID: 32454905 Free PMC article.
-
THEMIS-tery is solved.Nat Immunol. 2017 Mar 22;18(4):368-370. doi: 10.1038/ni.3708. Nat Immunol. 2017. PMID: 28323266 No abstract available.
References
-
- Acuto O, Di Bartolo V, Michel F. Tailoring T-cell receptor signals by proximal negative feedback mechanisms. Nat Rev Immunol. 2008;8:699–712. - PubMed
-
- Alon U. Network motifs: theory and experimental approaches. Nat Rev Genet. 2007;8:450–461. - PubMed
-
- Altman JD, Davis MM. MHC-peptide tetramers to visualize antigen-specific T cells. Curr Protoc Immunol. 2003 Chapter 17: Unit 17 13. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

