Oxidant exposure induces cysteine-rich protein 61 (CCN1) via c-Jun/AP-1 to reduce collagen expression in human dermal fibroblasts
- PMID: 25536346
- PMCID: PMC4275215
- DOI: 10.1371/journal.pone.0115402
Oxidant exposure induces cysteine-rich protein 61 (CCN1) via c-Jun/AP-1 to reduce collagen expression in human dermal fibroblasts
Abstract
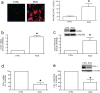
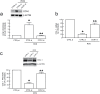
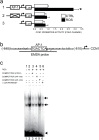
Human skin is a primary target of oxidative stress from reactive oxygen species (ROS) generated from both extrinsic and intrinsic sources. Oxidative stress inhibits the production of collagen, the most abundant protein in skin, and thus contributes to connective tissue aging. Here we report that cysteine-rich protein 61 (CCN1), a negative regulator of collagen production, is markedly induced by ROS and mediates loss of type I collagen in human dermal fibroblasts. Conversely, antioxidant N-acetyl-L-cysteine significantly reduced CCN1 expression and prevented ROS-induced loss of type I collagen in both human dermal fibroblasts and human skin in vivo. ROS increased c-Jun, a critical member of transcription factor AP-1 complex, and increased c-Jun binding to the AP-1 site of the CCN1 promoter. Functional blocking of c-Jun significantly reduced CCN1 promoter and gene expression and thus prevented ROS-induced loss of type I collagen. Targeting the c-Jun/CCN1 axis may provide clinical benefit for connective tissue aging in human skin.
Conflict of interest statement
Figures



References
-
- Droge W (2002) Free radicals in the physiological control of cell function. Physiol Rev 82:47–95. - PubMed
-
- Scharffetter-Kochanek K, Brenneisen P, Wenk J, Herrmann G, Ma W, et al. (2000) Photoaging of the skin from phenotype to mechanisms. Exp Gerontol 35:307–316. - PubMed
-
- Yaar M, Gilchrest BA (2007) Photoageing: mechanism, prevention and therapy. Br J Dermatol 157:874–887. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

