Rescue Treatment with L-Citrulline Inhibits Hypoxia-Induced Pulmonary Hypertension in Newborn Pigs
- PMID: 25536367
- PMCID: PMC4566047
- DOI: 10.1165/rcmb.2014-0351OC
Rescue Treatment with L-Citrulline Inhibits Hypoxia-Induced Pulmonary Hypertension in Newborn Pigs
Abstract
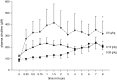
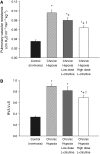
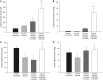
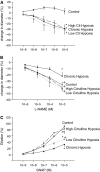
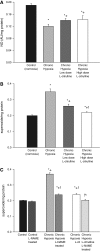
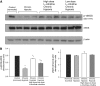
Infants with cardiopulmonary disorders associated with hypoxia develop pulmonary hypertension. We previously showed that initiation of oral L-citrulline before and continued throughout hypoxic exposure improves nitric oxide (NO) production and ameliorates pulmonary hypertension in newborn piglets. Rescue treatments, initiated after the onset of pulmonary hypertension, better approximate clinical strategies. Mechanisms by which L-citrulline improves NO production merit elucidation. The objective of this study was to determine whether starting L-citrulline after the onset of pulmonary hypertension inhibits disease progression and improves NO production by recoupling endothelial NO synthase (eNOS). Hypoxic and normoxic (control) piglets were studied. Some hypoxic piglets received oral L-citrulline starting on Day 3 of hypoxia and continuing throughout the remaining 7 days of hypoxic exposure. Catheters were placed for hemodynamic measurements, and pulmonary arteries were dissected to assess NO production and eNOS dimer-to-monomer ratios (a measure of eNOS coupling). Pulmonary vascular resistance was lower in L-citrulline-treated hypoxic piglets than in untreated hypoxic piglets but was higher than in normoxic controls. NO production and eNOS dimer-to-monomer ratios were greater in pulmonary arteries from L-citrulline-treated than from untreated hypoxic animals but were lower than in normoxic controls. When started after disease onset, oral L-citrulline treatment improves NO production by recoupling eNOS and inhibits the further development of chronic hypoxia-induced pulmonary hypertension in newborn piglets. Oral L-citrulline may be a novel strategy to halt or reverse pulmonary hypertension in infants suffering from cardiopulmonary conditions associated with hypoxia.
Keywords: nitric oxide signaling; superoxide; uncoupled eNOS.
Figures

References
-
- Mourani PM, Abman SH. Pulmonary vascular disease in bronchopulmonary dysplasia: pulmonary hypertension and beyond. Curr Opin Pediatr. 2013;25:329–337. - PubMed
-
- Stenmark KR, Abman SH. Lung vascular development: implications for the pathogenesis of bronchopulmonary dysplasia. Annu Rev Physiol. 2005;67:623–661. - PubMed
-
- Khemani E, McElhinney DB, Rhein L, Andrade O, Lacro RV, Thomas KC, Mullen MP. Pulmonary artery hypertension in formerly premature infants with bronchopulmonary dysplasia: clinical features and outcomes in the surfactant era. Pediatrics. 2007;120:1260–1269. - PubMed
-
- Fike CD, Aschner JL, Zhang Y, Kaplowitz MR. Impaired NO signaling in small pulmonary arteries of chronically hypoxic newborn piglets. Am J Physiol Lung Cell Mol Physiol. 2004;286:L1244–L1254. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

