Expression of Clock genes in the pineal glands of newborn rats with hypoxic-ischemic encephalopathy
- PMID: 25538743
- PMCID: PMC4268722
- DOI: 10.3969/j.issn.1673-5374.2012.028.008
Expression of Clock genes in the pineal glands of newborn rats with hypoxic-ischemic encephalopathy
Abstract
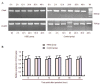
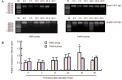
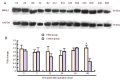
Clock genes are involved in circadian rhythm regulation, and surviving newborns with hypoxic-ischemic encephalopathy may present with sleep-wake cycle reversal. This study aimed to determine the expression of the clock genes Clock and Bmal1, in the pineal gland of rats with hypoxic-ischemic brain damage. Results showed that levels of Clock mRNA were not significantly changed within 48 hours after cerebral hypoxia and ischemia. Expression levels of CLOCK and BMAL1 protein were significantly higher after 48 hours. The levels of Bmal1 mRNA reached a peak at 36 hours, but were significantly reduced at 48 hours. Experimental findings indicate that Clock and Bmal1 genes were indeed expressed in the pineal glands of neonatal rats. At the initial stage (within 36 hours) of hypoxic-ischemic brain damage, only slight changes in the expression levels of these two genes were detected, followed by significant changes at 36-48 hours. These changes may be associated with circadian rhythm disorder induced by hypoxic-ischemic brain damage.
Keywords: Bmal1; Clock; brain; brain hypoxia; cerebral ischemia; mRNA; neonatal rats; neural regeneration; pineal gland; protein.
Conflict of interest statement
Figures

References
-
- de Vries LS, Jongmans MJ. Long-term outcome after neonatal hypoxic-ischaemic encephalopathy. Arch Dis Child Fetal Neonatal Ed. 2010;95(3):F220–224. - PubMed
-
- Perlman M, Shah PS. Hypoxic-ischemic encephalopathy: challenges in outcome and prediction. J Pediatr. 2011;158(2 Suppl):e51–54. - PubMed
-
- Kommedal S, Bódis G, Matkovits A, et al. Expression pattern of clock under acute phase-delay of the light/dark cycle in the chicken pineal model. Gen Comp Endocrinol. 2011;172(1):170–172. - PubMed
-
- Okano T, Yamamoto K, Okano K, et al. Chicken pineal clock genes: implication of BMAL2 as a bidirectional regulator in circadian clock oscillation. Genes Cells. 2001;6(9):825–836. - PubMed
-
- Shearman LP, Sriram S, Weaver DR, et al. Interacting molecular loops in the mammalian circadian clock. Science. 2000;288 (5468):1013–1019. - PubMed
LinkOut - more resources
Full Text Sources
