Carrier-mediated cocaine transport at the blood-brain barrier as a putative mechanism in addiction liability
- PMID: 25539501
- PMCID: PMC4368859
- DOI: 10.1093/ijnp/pyu001
Carrier-mediated cocaine transport at the blood-brain barrier as a putative mechanism in addiction liability
Erratum in
-
Erratum.Int J Neuropsychopharmacol. 2016 Apr 27;19(10):pyw031. doi: 10.1093/ijnp/pyw031. Int J Neuropsychopharmacol. 2016. PMID: 27207904 Free PMC article. No abstract available.
Abstract
Background: The rate of entry of cocaine into the brain is a critical factor that influences neuronal plasticity and the development of cocaine addiction. Until now, passive diffusion has been considered the unique mechanism known by which cocaine crosses the blood-brain barrier.
Methods: We reassessed mechanisms of transport of cocaine at the blood-brain barrier using a human cerebral capillary endothelial cell line (hCMEC/D3) and in situ mouse carotid perfusion.
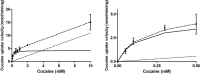
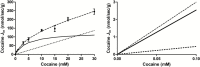
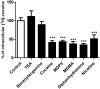
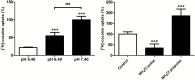
Results: Both in vivo and in vitro cocaine transport studies demonstrated the coexistence of a carrier-mediated process with passive diffusion. At pharmacological exposure level, passive diffusion of cocaine accounted for only 22.5% of the total cocaine influx in mice and 5.9% in hCMEC/D3 cells, whereas the carrier-mediated influx rate was 3.4 times greater than its passive diffusion rate in vivo. The functional identification of this carrier-mediated transport demonstrated the involvement of a proton antiporter that shared the properties of the previously characterized clonidine and nicotine transporter. The functionnal characterization suggests that the solute carrier (SLC) transporters Oct (Slc22a1-3), Mate (Slc47a1) and Octn (Slc22a4-5) are not involved in the cocaine transport in vivo and in vitro. Diphenhydramine, heroin, tramadol, cocaethylene, and norcocaine all strongly inhibited cocaine transport, unlike benzoylecgonine. Trans-stimulation studies indicated that diphenhydramine, nicotine, 3,4-methylenedioxyamphetamine (ecstasy) and the cathinone compound 3,4-methylenedioxypyrovalerone (MDPV) were also substrates of the cocaine transporter.
Conclusions: Cocaine transport at the BBB involves a proton-antiporter flux that is quantitatively much more important than its passive diffusion. The molecular identification and characterization of this transporter will provide new tools to understand its role in addictive mechanisms.
Keywords: biological transport; blood-brain barrier; cocaine; drug of abuse; pharmacokinetics..
© The Author 2014. Published by Oxford University Press. All rights reserved. For Permissions, please e-mail: journals.permissions@oup.com.
Figures
References
-
- Abbott NJ, Patabendige AA, Dolman DE, Yusof SR, Begley DJ. (2010). Structure and function of the blood-brain barrier. Neurobiol Dis 37:13–25. - PubMed
-
- Abreu ME, Bigelow GE, Fleisher L, Walsh SL. (2001). Effect of intravenous injection speed on responses to cocaine and hydromorphone in humans. Psychopharmacology (Berl) 154:76–84. - PubMed
-
- Agarwal S, Uchida Y, Mittapalli RK, Sane R, Terasaki T, Elmquist WF. (2012). Quantitative proteomics of transporter expression in brain capillary endothelial cells isolated from P-glycoprotein (P-gp), breast cancer resistance protein (Bcrp), and P-gp/Bcrp knockout mice. Drug Metab Dispos 40:1164–1169. - PMC - PubMed
-
- Andre P, Debray M, Scherrmann JM, Cisternino S. (2009). Clonidine transport at the mouse blood-brain barrier by a new H+ antiporter that interacts with addictive drugs. J Cereb Blood Flow Metab 29:1293–1304. - PubMed
-
- Andre P, Saubamea B, Cochois-Guegan V, Marie-Claire C, Cattelotte J, Smirnova M, Schinkel AH, Scherrmann JM, Cisternino S. (2012). Transport of biogenic amine neurotransmitters at the mouse blood-retina and blood-brain barriers by uptake1 and uptake2. J Cereb Blood Flow Metab 32:1989–2001. - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

