ETS transcription factor ETV2 directly converts human fibroblasts into functional endothelial cells
- PMID: 25540418
- PMCID: PMC4291653
- DOI: 10.1073/pnas.1413234112
ETS transcription factor ETV2 directly converts human fibroblasts into functional endothelial cells
Abstract
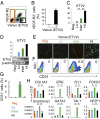
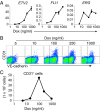
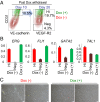
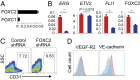
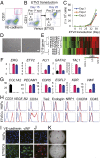
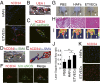
Transplantation of endothelial cells (ECs) is a promising therapeutic approach for ischemic disorders. In addition, the generation of ECs has become increasingly important for providing vascular plexus to regenerated organs, such as the liver. Although many attempts have been made to generate ECs from pluripotent stem cells and nonvascular cells, the minimum number of transcription factors that specialize in directly inducing vascular ECs remains undefined. Here, by screening 18 transcription factors that are important for both endothelial and hematopoietic development, we demonstrate that ets variant 2 (ETV2) alone directly converts primary human adult skin fibroblasts into functional vascular endothelial cells (ETVECs). In coordination with endogenous FOXC2 in fibroblasts, transduced ETV2 elicits expression of multiple key endothelial development factors, including FLI1, ERG, and TAL1, and induces expression of endothelial functional molecules, including EGFL7 and von Willebrand factor. Consequently, ETVECs exhibits EC characteristics in vitro and forms mature functional vasculature in Matrigel plugs transplanted in NOD SCID mice. Furthermore, ETVECs significantly improve blood flow recovery in a hind limb ischemic model using BALB/c-nu mice. Our study indicates that the creation of ETVECs provides further understanding of human EC development induced by ETV2.
Keywords: ETV2; angiogenesis; direct conversion; endothelial cells.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

