Profilin-1 serves as a gatekeeper for actin assembly by Arp2/3-dependent and -independent pathways
- PMID: 25543281
- PMCID: PMC4296256
- DOI: 10.1016/j.devcel.2014.10.026
Profilin-1 serves as a gatekeeper for actin assembly by Arp2/3-dependent and -independent pathways
Abstract
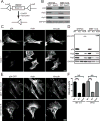
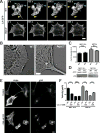
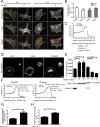
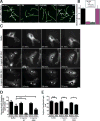
Cells contain multiple F-actin assembly pathways, including the Arp2/3 complex, formins, and Ena/VASP, which have largely been analyzed separately. They collectively generate the bulk of F-actin from a common pool of G-actin; however, the interplay and/or competition between these pathways remains poorly understood. Using fibroblast lines derived from an Arpc2 conditional knockout mouse, we established matched-pair cells with and without the Arp2/3 complex. Arpc2(-/-) cells lack lamellipodia and migrate more slowly than WT cells but have F-actin levels indistinguishable from controls. Actin assembly in Arpc2(-/-) cells was resistant to cytochalasin-D and was highly dependent on profilin-1 and Ena/VASP but not formins. Profilin-1 depletion in WT cells increased F-actin and Arp2/3 complex in lamellipodia. Conversely, addition of exogenous profilin-1 inhibited Arp2/3 complex actin nucleation in vitro and in vivo. Antagonism of the Arp2/3 complex by profilin-1 in cells appears to maintain actin homeostasis by balancing Arp2/3 complex-dependent and -independent actin assembly pathways.
Copyright © 2015 Elsevier Inc. All rights reserved.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

Comment in
-
Global resource distribution: allocation of actin building blocks by profilin.Dev Cell. 2015 Jan 12;32(1):5-6. doi: 10.1016/j.devcel.2014.12.022. Dev Cell. 2015. PMID: 25584793
References
-
- Bear J, Loureiro J, Libova I, Fassler R, Wehland J, Gertler F. Negative regulation of fibroblast motility by Ena/VASP proteins. Cell. 2000;101:717–728. - PubMed
-
- Bear JE, Svitkina TM, Krause M, Schafer DA, Loureiro JJ, Strasser GA, Maly IV, Chaga OY, Cooper JA, Borisy GG, et al. Antagonism between Ena/VASP Proteins and Actin Filament Capping Regulates Fibroblast Motility. Cell. 2002;109:509–521. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

