Insulin-like growth factor I reduces lipid oxidation and foam cell formation via downregulation of 12/15-lipoxygenase
- PMID: 25549319
- PMCID: PMC4298481
- DOI: 10.1016/j.atherosclerosis.2014.12.024
Insulin-like growth factor I reduces lipid oxidation and foam cell formation via downregulation of 12/15-lipoxygenase
Abstract
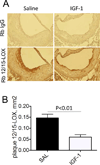
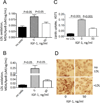
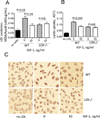
Objective: We have shown that insulin-like growth factor I (IGF-1) infusion in Apoe(-/-) mice decreased atherosclerotic plaque size and plaque macrophage and lipid content suggesting that IGF-1 suppressed formation of macrophage-derived foam cells. Since 12/15-lipoxygenase (12/15-LOX) plays an important role in OxLDL and foam cell formation, we hypothesized that IGF-1 downregulates 12/15-LOX, thereby suppressing lipid oxidation and foam cell formation.
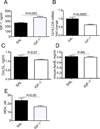
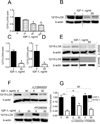
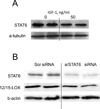
Approach and results: We found that IGF-1 decreased 12/15-LOX plaque immunopositivity and serum OxLDL levels in Apoe(-/-) mice. IGF-1 reduced 12/15-LOX protein and mRNA levels in cultured THP-1 macrophages and IGF-1 also decreased expression of STAT6 transcription factor. IGF-1 reduction in macrophage 12/15-LOX was mediated in part via a PI3 kinase- and STAT6-dependent transcriptional mechanism. IGF-1 suppressed THP-1 macrophage ability to oxidize lipids and form foam cells. IGF-1 downregulated 12/15-LOX in human blood-derived primary macrophages and IGF-1 decreased LDL oxidation induced by these cells. IGF-1 reduced LDL oxidation and formation of foam cells by wild type murine peritoneal macrophages, however these effects were completely blocked in 12/15-LOX-null macrophages suggesting that the ability of IGF-1 to reduce LDL oxidation and foam cells formation is dependent on its ability to downregulate 12/15-LOX.
Conclusions: Overall our data demonstrate that IGF-1 reduces lipid oxidation and foam cell formation via downregulation of 12/15-LOX and this mechanism may play a major role in the anti-atherosclerotic effects of IGF-1.
Keywords: Atherosclerosis; Foam cells; LDL/Oxidation/antioxidants; Lipoxygenase; Macrophages/monocytes; Oxidized lipids.
Published by Elsevier Ireland Ltd.
Figures
Similar articles
-
Inhibition of Orai1 Store-Operated Calcium Channel Prevents Foam Cell Formation and Atherosclerosis.Arterioscler Thromb Vasc Biol. 2016 Apr;36(4):618-28. doi: 10.1161/ATVBAHA.116.307344. Epub 2016 Feb 25. Arterioscler Thromb Vasc Biol. 2016. PMID: 26916730
-
Critical role of macrophage 12/15-lipoxygenase for atherosclerosis in apolipoprotein E-deficient mice.Circulation. 2004 Oct 5;110(14):2024-31. doi: 10.1161/01.CIR.0000143628.37680.F6. Epub 2004 Sep 27. Circulation. 2004. PMID: 15451785
-
Insulin-Like Growth Factor-1 Receptor Deficiency in Macrophages Accelerates Atherosclerosis and Induces an Unstable Plaque Phenotype in Apolipoprotein E-Deficient Mice.Circulation. 2016 Jun 7;133(23):2263-78. doi: 10.1161/CIRCULATIONAHA.116.021805. Epub 2016 May 6. Circulation. 2016. PMID: 27154724 Free PMC article.
-
Lipoxygenases and atherosclerosis: protection versus pathogenesis.Free Radic Biol Med. 2000 Jun 15;28(12):1726-34. doi: 10.1016/s0891-5849(00)00230-6. Free Radic Biol Med. 2000. PMID: 10946214 Review.
-
LOX-1, OxLDL, and atherosclerosis.Mediators Inflamm. 2013;2013:152786. doi: 10.1155/2013/152786. Epub 2013 Jul 10. Mediators Inflamm. 2013. PMID: 23935243 Free PMC article. Review.
Cited by
-
Ox-LDL Upregulates IL-6 Expression by Enhancing NF-κB in an IGF2-Dependent Manner in THP-1 Macrophages.Inflammation. 2015 Dec;38(6):2116-23. doi: 10.1007/s10753-015-0194-1. Inflammation. 2015. PMID: 26063187
-
IGF-1 and cardiovascular disease.Growth Horm IGF Res. 2019 Apr;45:6-16. doi: 10.1016/j.ghir.2019.01.002. Epub 2019 Jan 31. Growth Horm IGF Res. 2019. PMID: 30735831 Free PMC article. Review.
-
Construction and Analysis of lncRNA-Associated ceRNA Network in Atherosclerotic Plaque Formation.Biomed Res Int. 2022 Apr 15;2022:4895611. doi: 10.1155/2022/4895611. eCollection 2022. Biomed Res Int. 2022. PMID: 35463977 Free PMC article.
-
Serum paraoxonase level and paraoxonase polymorphism in patients with acromegaly.Redox Rep. 2016 Nov;21(6):281-6. doi: 10.1080/13510002.2015.1133036. Epub 2016 Feb 15. Redox Rep. 2016. PMID: 26863932 Free PMC article.
-
Whole Body Vibration Retards Progression of Atherosclerosis via Insulin-Like Growth Factor 1 in Apolipoprotein E-Deficient Mice.Biomed Res Int. 2018 Mar 7;2018:4934861. doi: 10.1155/2018/4934861. eCollection 2018. Biomed Res Int. 2018. PMID: 29707570 Free PMC article.
References
-
- Parthasarathy S, Steinberg D, Witztum JL. The role of oxidized low-density lipoproteins in the pathogenesis of atherosclerosis. Annu Rev Med. 1992;43:219–225. - PubMed
-
- Yu XH, et al. Foam cells in atherosclerosis. Clin Chim Acta. 2013;424:245–252. - PubMed
-
- Funk CD. Lipoxygenase pathways as mediators of early inflammatory events in atherosclerosis. Arterioscler Thromb Vasc Biol. 2006;26(6):1204–1206. - PubMed
-
- Cyrus T, et al. Absence of 12/15-lipoxygenase expression decreases lipid peroxidation and atherogenesis in apolipoprotein e-deficient mice. Circulation. 2001;103(18):2277–2282. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

