Inorganic Janus particles for biomedical applications
- PMID: 25551063
- PMCID: PMC4273258
- DOI: 10.3762/bjnano.5.244
Inorganic Janus particles for biomedical applications
Abstract
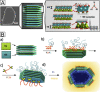
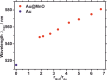
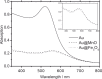
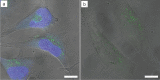
Based on recent developments regarding the synthesis and design of Janus nanoparticles, they have attracted increased scientific interest due to their outstanding properties. There are several combinations of multicomponent hetero-nanostructures including either purely organic or inorganic, as well as composite organic-inorganic compounds. Janus particles are interconnected by solid state interfaces and, therefore, are distinguished by two physically or chemically distinct surfaces. They may be, for instance, hydrophilic on one side and hydrophobic on the other, thus, creating giant amphiphiles revealing the endeavor of self-assembly. Novel optical, electronic, magnetic, and superficial properties emerge in inorganic Janus particles from their dimensions and unique morphology at the nanoscale. As a result, inorganic Janus nanoparticles are highly versatile nanomaterials with great potential in different scientific and technological fields. In this paper, we highlight some advances in the synthesis of inorganic Janus nanoparticles, focusing on the heterogeneous nucleation technique and characteristics of the resulting high quality nanoparticles. The properties emphasized in this review range from the monodispersity and size-tunability and, therefore, precise control over size-dependent features, to the biomedical application as theranostic agents. Hence, we show their optical properties based on plasmonic resonance, the two-photon activity, the magnetic properties, as well as their biocompatibility and interaction with human blood serum.
Keywords: Janus particles; MRI; Multi-photon); bioimaging (CT; hetero-nanoparticles; protein corona; synthesis.
Figures












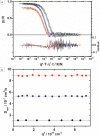
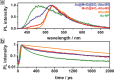
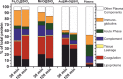
References
-
- Ozin G A. Adv Mater. 1992;4:612–649. doi: 10.1002/adma.19920041003. - DOI
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
