Intramolecular N-glycan/polypeptide interactions observed at multiple N-glycan remodeling steps through [(13)C,(15)N]-N-acetylglucosamine labeling of immunoglobulin G1
- PMID: 25551295
- PMCID: PMC4302832
- DOI: 10.1021/bi501380t
Intramolecular N-glycan/polypeptide interactions observed at multiple N-glycan remodeling steps through [(13)C,(15)N]-N-acetylglucosamine labeling of immunoglobulin G1
Abstract
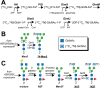
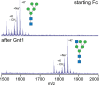
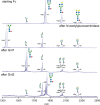
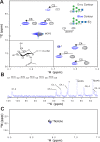
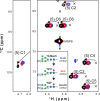
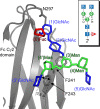
Asparagine-linked (N) glycosylation is a common eukaryotic protein modification that affects protein folding, function, and stability through intramolecular interactions between N-glycan and polypeptide residues. Attempts to characterize the structure-activity relationship of each N-glycan are hindered by inherent properties of the glycoprotein, including glycan conformational and compositional heterogeneity. These limitations can be addressed by using a combination of nuclear magnetic resonance techniques following enzymatic glycan remodeling to simultaneously generate homogeneous glycoforms. However, widely applicable methods do not yet exist. To address this technological gap, immature glycoforms of the immunoglobulin G1 fragment crystallizable (Fc) were isolated in a homogeneous state and enzymatically remodeled with [(13)C,(15)N]-N-acetylglucosamine (GlcNAc). UDP-[(13)C,(15)N]GlcNAc was synthesized enzymatically in a one-pot reaction from [(13)C]glucose and [(15)N-amido]glutamine. Modifying Fc with recombinantly expressed glycosyltransferases (Gnt1 and Gnt2) and UDP-[(13)C,(15)N]GlcNAc resulted in complete glycoform conversion as judged by mass spectrometry. Two-dimensional heteronuclear single-quantum coherence spectra of the Gnt1 product, containing a single [(13)C,(15)N]GlcNAc residue on each N-glycan, showed that the N-glycan is stabilized through interactions with polypeptide residues. Similar spectra of homogeneous glycoforms, halted at different points along the N-glycan remodeling pathway, revealed the presence of an increased level of interaction between the N-glycan and polypeptide at each step, including mannose trimming, as the N-glycan was converted to a complex-type, biantennary form. Thus, conformational restriction increases as Fc N-glycan maturation proceeds. Gnt1 and Gnt2 catalyze fundamental reactions in the synthesis of every glycoprotein with a complex-type N-glycan; thus, the strategies presented herein can be applied to a broad range of glycoprotein studies.
Figures


References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

