Spatiotemporal dynamics of optogenetically induced and spontaneous seizure transitions in primary generalized epilepsy
- PMID: 25552645
- PMCID: PMC4416582
- DOI: 10.1152/jn.01040.2014
Spatiotemporal dynamics of optogenetically induced and spontaneous seizure transitions in primary generalized epilepsy
Abstract
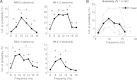
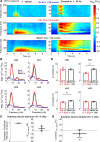
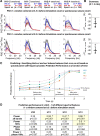
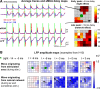
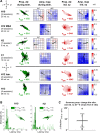
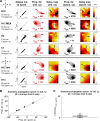
Transitions into primary generalized epileptic seizures occur abruptly and synchronously across the brain. Their potential triggers remain unknown. We used optogenetics to causally test the hypothesis that rhythmic population bursting of excitatory neurons in a local neocortical region can rapidly trigger absence seizures. Most previous studies have been purely correlational, and it remains unclear whether epileptiform events induced by rhythmic stimulation (e.g., sensory/electrical) mimic actual spontaneous seizures, especially regarding their spatiotemporal dynamics. In this study, we used a novel combination of intracortical optogenetic stimulation and microelectrode array recordings in freely moving WAG/Rij rats, a model of absence epilepsy with a cortical focus in the somatosensory cortex (SI). We report three main findings: 1) Brief rhythmic bursting, evoked by optical stimulation of neocortical excitatory neurons at frequencies around 10 Hz, induced seizures consisting of self-sustained spike-wave discharges (SWDs) for about 10% of stimulation trials. The probability of inducing seizures was frequency-dependent, reaching a maximum at 10 Hz. 2) Local field potential power before stimulation and response amplitudes during stimulation both predicted seizure induction, demonstrating a modulatory effect of brain states and neural excitation levels. 3) Evoked responses during stimulation propagated as cortical waves, likely reaching the cortical focus, which in turn generated self-sustained SWDs after stimulation was terminated. Importantly, SWDs during induced and spontaneous seizures propagated with the same spatiotemporal dynamics. Our findings demonstrate that local rhythmic bursting of excitatory neurons in neocortex at particular frequencies, under susceptible ongoing brain states, is sufficient to trigger primary generalized seizures with stereotypical spatiotemporal dynamics.
Keywords: absence seizures; epilepsy; microelectrode arrays; optogenetics.
Figures





References
-
- Benjamini Y, Hochberg Y. Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc Series B Stat Methodol 57: 289–300, 1995.
-
- Benjamini Y, Yekutieli D. The control of the false discovery rate in multiple testing under dependency. Ann Stat 29: 1165–1188, 2001.
-
- Bernier GP, Richer F, Giard N, Bouvier G, Mercier M, Turmel A, Saint-Hilaire JM. Electrical stimulation of the human brain in epilepsy. Epilepsia 31: 513-52, 1990. - PubMed
-
- Blume WT, Jones DC, Pathak P. Properties of after-discharges from cortical electrical stimulation in focal epilepsies. Clin Neurophysiol 115: 982–989, 2004. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

