Development and validation of the JAX Cancer Treatment Profile™ for detection of clinically actionable mutations in solid tumors
- PMID: 25562415
- PMCID: PMC4481190
- DOI: 10.1016/j.yexmp.2014.12.009
Development and validation of the JAX Cancer Treatment Profile™ for detection of clinically actionable mutations in solid tumors
Abstract
Background: The continued development of targeted therapeutics for cancer treatment has required the concomitant development of more expansive methods for the molecular profiling of the patient's tumor. We describe the validation of the JAX Cancer Treatment Profile™ (JAX-CTP™), a next generation sequencing (NGS)-based molecular diagnostic assay that detects actionable mutations in solid tumors to inform the selection of targeted therapeutics for cancer treatment.
Methods: NGS libraries are generated from DNA extracted from formalin fixed paraffin embedded tumors. Using hybrid capture, the genes of interest are enriched and sequenced on the Illumina HiSeq 2500 or MiSeq sequencers followed by variant detection and functional and clinical annotation for the generation of a clinical report.
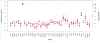
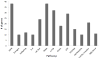
Results: The JAX-CTP™ detects actionable variants, in the form of single nucleotide variations and small insertions and deletions (≤50 bp) in 190 genes in specimens with a neoplastic cell content of ≥10%. The JAX-CTP™ is also validated for the detection of clinically actionable gene amplifications.
Conclusions: There is a lack of consensus in the molecular diagnostics field on the best method for the validation of NGS-based assays in oncology, thus the importance of communicating methods, as contained in this report. The growing number of targeted therapeutics and the complexity of the tumor genome necessitate continued development and refinement of advanced assays for tumor profiling to enable precision cancer treatment.
Keywords: Actionable; Cancer; FFPE; Mutation; Panel.
Copyright © 2014 Elsevier Inc. All rights reserved.
Figures


References
-
- Singh RR, Patel KP, Routbort MJ, Reddy NG, Barkoh BA, Handal B, Kanagal-Shamanna R, Greaves WO, Medeiros LJ, Aldape KD, Luthra R. Clinical validation of a next-generation sequencing screen for mutational hotspots in 46 cancer-related genes. The Journal of molecular diagnostics : JMD. 2013;15:607–22. - PubMed
-
- Johnson DB, Dahlman KH, Knol J, Gilbert J, Puzanov I, Means-Powell J, Balko JM, Lovly CM, Murphy BA, Goff LW, Abramson VG, Crispens MA, Mayer IA, Berlin JD, Horn L, Keedy VL, Reddy NM, Arteaga CL, Sosman JA, Pao W. Enabling a genetically informed approach to cancer medicine: a retrospective evaluation of the impact of comprehensive tumor profiling using a targeted next-generation sequencing panel. The oncologist. 2014;19:616–22. - PMC - PubMed
-
- Tran B, Brown AM, Bedard PL, Winquist E, Goss GD, Hotte SJ, Welch SA, Hirte HW, Zhang T, Stein LD, Ferretti V, Watt S, Jiao W, Ng K, Ghai S, Shaw P, Petrocelli T, Hudson TJ, Neel BG, Onetto N, Siu LL, McPherson JD, Kamel-Reid S, Dancey JE. Feasibility of real time next generation sequencing of cancer genes linked to drug response: results from a clinical trial. International journal of cancer Journal international du cancer. 2013;132:1547–55. - PubMed
-
- Said R, Hong DS, Warneke CL, Lee JJ, Wheler JJ, Janku F, Naing A, Falchook GS, Fu S, Piha-Paul S, Tsimberidou AM, Kurzrock R. P53 mutations in advanced cancers: clinical characteristics, outcomes, and correlation between progression-free survival and bevacizumab-containing therapy. Oncotarget. 2013;4:705–14. - PMC - PubMed
-
- Petrelli F, Coinu A, Cabiddu M, Ghilardi M, Barni S. KRAS as prognostic biomarker in metastatic colorectal cancer patients treated with bevacizumab: a pooled analysis of 12 published trials. Medical oncology. 2013;30:650. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

