Genetic erosion impedes adaptive responses to stressful environments
- PMID: 25568035
- PMCID: PMC3353342
- DOI: 10.1111/j.1752-4571.2011.00214.x
Genetic erosion impedes adaptive responses to stressful environments
Abstract
Biodiversity is increasingly subjected to human-induced changes of the environment. To persist, populations continually have to adapt to these often stressful changes including pollution and climate change. Genetic erosion in small populations, owing to fragmentation of natural habitats, is expected to obstruct such adaptive responses: (i) genetic drift will cause a decrease in the level of adaptive genetic variation, thereby limiting evolutionary responses; (ii) inbreeding and the concomitant inbreeding depression will reduce individual fitness and, consequently, the tolerance of populations to environmental stress. Importantly, inbreeding generally increases the sensitivity of a population to stress, thereby increasing the amount of inbreeding depression. As adaptation to stress is most often accompanied by increased mortality (cost of selection), the increase in the 'cost of inbreeding' under stress is expected to severely hamper evolutionary adaptive processes. Inbreeding thus plays a pivotal role in this process and is expected to limit the probability of genetically eroded populations to successfully adapt to stressful environmental conditions. Consequently, the dynamics of small fragmented populations may differ considerably from large nonfragmented populations. The resilience of fragmented populations to changing and deteriorating environments is expected to be greatly decreased. Alleviating inbreeding depression, therefore, is crucial to ensure population persistence.
Keywords: anthropogenic stress; changing environments; cost of inbreeding; genetic drift; genetic variation; habitat fragmentation; inbreeding depression; population persistence.
Figures
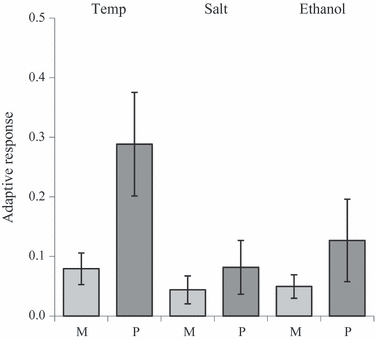
References
-
- Allendorf FW, Hohenlohe PA, Luikart G. Genomics and the future of conservation genetics. Nature Reviews Genetics. 2010;11:697–709. - PubMed
-
- Armbuster P, Reed DH. Inbreeding depression in benign and stressful environments. Heredity. 2005;95:235–242. - PubMed
-
- Bakker J, van Rijswijk MEC, Weissing FJ, Bijlsma R. Consequences of fragmentation for the ability to adapt to novel environments in experimental Drosophila metapopulations. Conservation Genetics. 2010;11:449–462.
LinkOut - more resources
Full Text Sources
Research Materials

