A chemiosmotic mechanism of symport
- PMID: 25568085
- PMCID: PMC4321259
- DOI: 10.1073/pnas.1419325112
A chemiosmotic mechanism of symport
Abstract
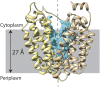
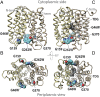
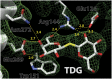
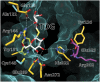
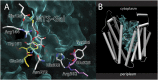
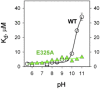
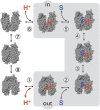
Lactose permease (LacY), a paradigm for the largest family of membrane transport proteins, catalyzes the coupled translocation of a galactoside and an H(+) across the Escherichia coli membrane (galactoside/H(+) symport). Initial X-ray structures reveal N- and C-terminal domains, each with six largely irregular transmembrane helices surrounding an aqueous cavity open to the cytoplasm. Recently, a structure with a narrow periplasmic opening and an occluded galactoside was obtained, confirming many observations and indicating that sugar binding involves induced fit. LacY catalyzes symport by an alternating access mechanism. Experimental findings garnered over 45 y indicate the following: (i) The limiting step for lactose/H(+) symport in the absence of the H(+) electrochemical gradient (∆µ̃H+) is deprotonation, whereas in the presence of ∆µ̃H+, the limiting step is opening of apo LacY on the other side of the membrane; (ii) LacY must be protonated to bind galactoside (the pK for binding is ∼10.5); (iii) galactoside binding and dissociation, not ∆µ̃H+, are the driving forces for alternating access; (iv) galactoside binding involves induced fit, causing transition to an occluded intermediate that undergoes alternating access; (v) galactoside dissociates, releasing the energy of binding; and (vi) Arg302 comes into proximity with protonated Glu325, causing deprotonation. Accumulation of galactoside against a concentration gradient does not involve a change in Kd for sugar on either side of the membrane, but the pKa (the affinity for H(+)) decreases markedly. Thus, transport is driven chemiosmotically but, contrary to expectation, ∆µ̃H+ acts kinetically to control the rate of the process.
Keywords: MFS; X-ray crystal structure; conformational change; membrane proteins; transport.
Conflict of interest statement
The author declares no conflict of interest.
Figures

References
-
- Madej MG, Kaback HR. 2014. The life and times of Lac permease: Crystals ain’t enough, but they certainly do help. Membrane Transporter Function: To Structure and Beyond, Springer Series in Biophysics: Transporters, eds Ziegler C, Kraemer R (Springer, Heidelberg) Vol 17, pp 121–158.
-
- Abramson J, et al. Structure and mechanism of the lactose permease of Escherichia coli. Science. 2003;301(5633):610–615. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

