Temporal plasticity involved in recovery from manual dexterity deficit after motor cortex lesion in macaque monkeys
- PMID: 25568105
- PMCID: PMC4287160
- DOI: 10.1523/JNEUROSCI.1737-14.2015
Temporal plasticity involved in recovery from manual dexterity deficit after motor cortex lesion in macaque monkeys
Abstract
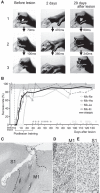
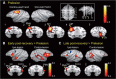
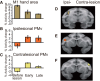
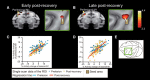
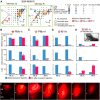
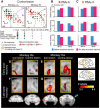
The question of how intensive motor training restores motor function after brain damage or stroke remains unresolved. Here we show that the ipsilesional ventral premotor cortex (PMv) and perilesional primary motor cortex (M1) of rhesus macaque monkeys are involved in the recovery of manual dexterity after a lesion of M1. A focal lesion of the hand digit area in M1 was made by means of ibotenic acid injection. This lesion initially caused flaccid paralysis in the contralateral hand but was followed by functional recovery of hand movements, including precision grip, during the course of daily postlesion motor training. Brain imaging of regional cerebral blood flow by means of H2 (15)O-positron emission tomography revealed enhanced activity of the PMv during the early postrecovery period and increased functional connectivity within M1 during the late postrecovery period. The causal role of these areas in motor recovery was confirmed by means of pharmacological inactivation by muscimol during the different recovery periods. These findings indicate that, in both the remaining primary motor and premotor cortical areas, time-dependent plastic changes in neural activity and connectivity are involved in functional recovery from the motor deficit caused by the M1 lesion. Therefore, it is likely that the PMv, an area distant from the core of the lesion, plays an important role during the early postrecovery period, whereas the perilesional M1 contributes to functional recovery especially during the late postrecovery period.
Keywords: brain activation; functional compensation; macaque monkey; precision grip; primate.
Copyright © 2015 the authors 0270-6474/15/350084-12$15.00/0.
Figures
Similar articles
-
Effects of motor training on the recovery of manual dexterity after primary motor cortex lesion in macaque monkeys.J Neurophysiol. 2008 Feb;99(2):773-86. doi: 10.1152/jn.01001.2007. Epub 2007 Dec 19. J Neurophysiol. 2008. PMID: 18094104
-
Asymmetric and Distant Effects of a Unilateral Lesion of the Primary Motor Cortex on the Bilateral Supplementary Motor Areas in Adult Macaque Monkeys.J Neurosci. 2018 Dec 12;38(50):10644-10656. doi: 10.1523/JNEUROSCI.0904-18.2018. Epub 2018 Oct 24. J Neurosci. 2018. PMID: 30355637 Free PMC article.
-
Role of primary motor cortex in the control of manual dexterity assessed via sequential bilateral lesion in the adult macaque monkey: A case study.Neuroscience. 2017 Aug 15;357:303-324. doi: 10.1016/j.neuroscience.2017.06.018. Epub 2017 Jun 17. Neuroscience. 2017. PMID: 28629845
-
Training-induced recovery of manual dexterity after a lesion in the motor cortex.Keio J Med. 2010;59(1):4-9. doi: 10.2302/kjm.59.4. Keio J Med. 2010. PMID: 20375652 Review.
-
Cortical and subcortical compensatory mechanisms after spinal cord injury in monkeys.Exp Neurol. 2012 May;235(1):152-61. doi: 10.1016/j.expneurol.2011.08.013. Epub 2011 Aug 23. Exp Neurol. 2012. PMID: 21884698 Review.
Cited by
-
Evaluating the Abnormality of Bilateral Motor Cortex Activity in Subacute Stroke Patients Executing a Unimanual Motor Task With Increasing Demand on Precision.Front Neurol. 2022 May 25;13:836716. doi: 10.3389/fneur.2022.836716. eCollection 2022. Front Neurol. 2022. PMID: 35693005 Free PMC article.
-
The ventral striatum contributes to the activity of the motor cortex and motor outputs in monkeys.Front Syst Neurosci. 2022 Aug 29;16:979272. doi: 10.3389/fnsys.2022.979272. eCollection 2022. Front Syst Neurosci. 2022. PMID: 36211590 Free PMC article.
-
Development and Characterization of a Macaque Model of Focal Internal Capsular Infarcts.PLoS One. 2016 May 5;11(5):e0154752. doi: 10.1371/journal.pone.0154752. eCollection 2016. PLoS One. 2016. PMID: 27149111 Free PMC article.
-
Neural Activity during Voluntary Movements in Each Body Representation of the Intracortical Microstimulation-Derived Map in the Macaque Motor Cortex.PLoS One. 2016 Aug 5;11(8):e0160720. doi: 10.1371/journal.pone.0160720. eCollection 2016. PLoS One. 2016. PMID: 27494282 Free PMC article.
-
Accounting for the valley of recovery during post-stroke rehabilitation training via a model-based analysis of macaque manual dexterity.Front Rehabil Sci. 2022 Dec 20;3:1042912. doi: 10.3389/fresc.2022.1042912. eCollection 2022. Front Rehabil Sci. 2022. PMID: 36644290 Free PMC article.
References
-
- Arikan R, Blake NM, Erinjeri JP, Woolsey TA, Giraud L, Highstein SM. A method to measure the effective spread of focally injected muscimol into the central nervous system with electrophysiology and light microscopy. J Neurosci Methods. 2002;118:51–57. doi: 10.1016/S0165-0270(02)00143-7. - DOI - PubMed
-
- Courtine G, Bunge MB, Fawcett JW, Grossman RG, Kaas JH, Lemon R, Maier I, Martin J, Nudo RJ, Ramon-Cueto A, Rouiller EM, Schnell L, Wannier T, Schwab ME, Edgerton VR. Can experiments in nonhuman primates expedite the translation of treatments for spinal cord injury in humans? Nat Med. 2007;13:561–566. doi: 10.1038/nm1595. - DOI - PMC - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
