Overweight in mice and enhanced adipogenesis in vitro are associated with lack of the hedgehog coreceptor boc
- PMID: 25576054
- PMCID: PMC4439556
- DOI: 10.2337/db14-1017
Overweight in mice and enhanced adipogenesis in vitro are associated with lack of the hedgehog coreceptor boc
Abstract
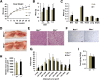
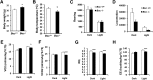
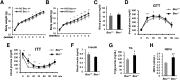
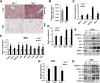
Obesity arises from a combination of genetic, environmental, and behavioral factors. However, the processes that regulate white adipose tissue (WAT) expansion at the level of the adipocyte are not well understood. The Hedgehog (HH) pathway plays a conserved role in adipogenesis, inhibiting fat formation in vivo and in vitro, but it has not been shown that mice with reduced HH pathway activity have enhanced adiposity. We report that mice lacking the HH coreceptor BOC displayed age-related overweight and excess WAT. They also displayed alterations in some metabolic parameters but normal food intake. Furthermore, they had an exacerbated response to a high-fat diet, including enhanced weight gain and adipocyte hypertrophy, livers with greater fat accumulation, and elevated expression of genes related to adipogenesis, lipid metabolism, and adipokine production. Cultured Boc(-/-) mouse embryo fibroblasts showed enhanced adipogenesis relative to Boc(+/+) cells, and they expressed reduced levels of HH pathway target genes. Therefore, a loss-of-function mutation in an HH pathway component is associated with WAT accumulation and overweight in mice. Variant alleles of such HH regulators may contribute to WAT accumulation in human individuals with additional genetic or lifestyle-based predisposition to obesity.
© 2015 by the American Diabetes Association. Readers may use this article as long as the work is properly cited, the use is educational and not for profit, and the work is not altered.
Figures



Similar articles
-
Differential screening identifies transcripts with depot-dependent expression in white adipose tissues.BMC Genomics. 2008 Aug 22;9:397. doi: 10.1186/1471-2164-9-397. BMC Genomics. 2008. PMID: 18721461 Free PMC article.
-
GRK5 deficiency decreases diet-induced obesity and adipogenesis.Biochem Biophys Res Commun. 2012 May 4;421(2):312-7. doi: 10.1016/j.bbrc.2012.04.006. Epub 2012 Apr 7. Biochem Biophys Res Commun. 2012. PMID: 22507984
-
Hedgehog signaling pathway regulated the target genes for adipogenesis in silkworm Bombyx mori.Insect Sci. 2015 Oct;22(5):587-96. doi: 10.1111/1744-7917.12164. Epub 2014 Oct 28. Insect Sci. 2015. PMID: 25154865
-
Reduced white fat mass in adult mice bearing a truncated Patched 1.Int J Biol Sci. 2008 Jan 26;4(1):29-36. doi: 10.7150/ijbs.4.29. Int J Biol Sci. 2008. PMID: 18274621 Free PMC article.
-
Cdon and Boc: Two transmembrane proteins implicated in cell-cell communication.Int J Biochem Cell Biol. 2012 May;44(5):698-702. doi: 10.1016/j.biocel.2012.01.019. Epub 2012 Feb 3. Int J Biochem Cell Biol. 2012. PMID: 22326621 Review.
Cited by
-
Impact of obese levels on the hepatic expression of nuclear receptors and drug-metabolizing enzymes in adult and offspring mice.Acta Pharm Sin B. 2020 Jan;10(1):171-185. doi: 10.1016/j.apsb.2019.10.009. Epub 2019 Nov 28. Acta Pharm Sin B. 2020. PMID: 31993314 Free PMC article.
-
Exercise reduces metabolic burden while altering the immune system in aged mice.Aging (Albany NY). 2021 Jan 6;13(1):1294-1313. doi: 10.18632/aging.202312. Epub 2021 Jan 6. Aging (Albany NY). 2021. PMID: 33406502 Free PMC article.
-
Human germline hedgehog pathway mutations predispose to fatty liver.J Hepatol. 2017 Oct;67(4):809-817. doi: 10.1016/j.jhep.2017.06.008. Epub 2017 Jun 21. J Hepatol. 2017. PMID: 28645738 Free PMC article.
-
Endothelial PRMT7 prevents dysfunction, promotes revascularization and enhances cardiac recovery post-myocardial infarction.Exp Mol Med. 2025 Aug 5. doi: 10.1038/s12276-025-01517-x. Online ahead of print. Exp Mol Med. 2025. PMID: 40764455
-
Cdon deficiency causes cardiac remodeling through hyperactivation of WNT/β-catenin signaling.Proc Natl Acad Sci U S A. 2017 Feb 21;114(8):E1345-E1354. doi: 10.1073/pnas.1615105114. Epub 2017 Feb 2. Proc Natl Acad Sci U S A. 2017. PMID: 28154134 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

