Trends in oral anticoagulant choice for acute stroke patients with nonvalvular atrial fibrillation in Japan: the SAMURAI-NVAF study
- PMID: 25581108
- PMCID: PMC4964913
- DOI: 10.1111/ijs.12452
Trends in oral anticoagulant choice for acute stroke patients with nonvalvular atrial fibrillation in Japan: the SAMURAI-NVAF study
Abstract
Background: Large clinical trials are lack of data on non-vitamin K antagonist oral anticoagulants for acute stroke patients.
Aim: To evaluate the choice of oral anticoagulants at acute hospital discharge in stroke patients with nonvalvular atrial fibrillation and clarify the underlying characteristics potentially affecting that choice using the multicenter Stroke Acute Management with Urgent Risk-factor Assessment and Improvement-NVAF registry (ClinicalTrials.gov NCT01581502).
Method: The study included 1192 acute ischemic stroke/transient ischemic attack patients with nonvalvular atrial fibrillation (527 women, 77·7 ± 9·9 years old) between September 2011 and March 2014, during which three nonvitamin K antagonist oral anticoagulant oral anticoagulants were approved for clinical use. Oral anticoagulant choice at hospital discharge (median 23-day stay) was assessed.
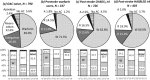
Results: Warfarin was chosen for 650 patients, dabigatran for 203, rivaroxaban for 238, and apixaban for 25. Over the three 10-month observation periods, patients taking warfarin gradually decreased to 46·5% and those taking nonvitamin K antagonist oral anticoagulants increased to 48·0%. As compared with warfarin users, patients taking nonvitamin K antagonist oral anticoagulants included more men, were younger, more frequently had small infarcts, and had lower scores for poststroke CHADS2 , CHA2 DS2 -VASc, and HAS-BLED, admission National Institutes of Health stroke scale, and discharge modified Rankin Scale. Nonvitamin K antagonist oral anticoagulants were started at a median of four-days after stroke onset without early intracranial hemorrhage. Patients starting nonvitamin K antagonist oral anticoagulants earlier had smaller infarcts and lower scores for the admission National Institutes of Health stroke scale and the discharge modified Rankin Scale than those starting later. Choice of nonvitamin K antagonist oral anticoagulants was independently associated with 20-day or shorter hospitalization (OR 2·46, 95% CI 1·87-3·24).
Conclusions: Warfarin use at acute hospital discharge was still common in the initial years after approval of nonvitamin K antagonist oral anticoagulants, although nonvitamin K antagonist oral anticoagulant users increased gradually. The index stroke was milder and ischemia-risk indices were lower in nonvitamin K antagonist oral anticoagulant users than in warfarin users. Early initiation of nonvitamin K antagonist oral anticoagulants seemed safe.
Keywords: acute stroke care; anticoagulation; atrial fibrillation; embolism; prevention.
© 2015 The Authors. International Journal of Stroke published by John Wiley & Sons Ltd on behalf of World Stroke Organization.
Figures


References
-
- Husted S, de Caterina R, Andreotti F et al Non‐vitamin K antagonist oral anticoagulants (NOACs): no longer new or novel. Thromb Haemost 2014; 111:781–782. - PubMed
-
- Connoly SJ, Ezekowitz MD, Yusuf S et al Dabigatran versus warfarin in patients with atrial fibrillation. N Engl J Med 2009; 361:1139–1151. - PubMed
-
- Patel MR, Mahaffey KW, Garg J et al Rivaroxaban versus warfarin in nonvalvular atrial fibrillation. N Engl J Med 2011; 365:883–891. - PubMed
-
- Granger CB, Alexander JH, McMurray JJ et al Apixaban versus warfarin in patients with atrial fibrillation. N Engl J Med 2011; 365:981–992. - PubMed
-
- Giugliano RP, Ruff CT, Braunwald E et al Edoxaban versus warfarin in patients with atrial fibrillation. N Engl J Med 2013; 369:2093–2104. - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

