Ricin trafficking in cells
- PMID: 25584427
- PMCID: PMC4303813
- DOI: 10.3390/toxins7010049
Ricin trafficking in cells
Abstract
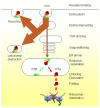
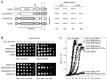
The heterodimeric plant toxin ricin binds exposed galactosyls at the cell surface of target mammalian cells, and, following endocytosis, is transported in vesicular carriers to the endoplasmic reticulum (ER). Subsequently, the cell-binding B chain (RTB) and the catalytic A chain (RTA) are separated reductively, RTA embeds in the ER membrane and then retrotranslocates (or dislocates) across this membrane. The protein conducting channels used by RTA are usually regarded as part of the ER-associated protein degradation system (ERAD) that removes misfolded proteins from the ER for destruction by the cytosolic proteasomes. However, unlike ERAD substrates, cytosolic RTA avoids destruction and folds into a catalytic conformation that inactivates its target ribosomes. Protein synthesis ceases, and subsequently the cells die apoptotically. This raises questions about how this protein avoids the pathways that are normally sanctioned for ER-dislocating substrates. In this review we focus on the molecular events that occur with non-tagged ricin and its isolated subunits at the ER-cytosol interface. This focus reveals that intra-membrane interactions of RTA may control its fate, an area that warrants further investigation.
Figures





References
-
- Sandvig K., Olsnes S., Pihl A. Kinetics of binding of the toxic lectins abrin and ricin to surface receptors of human cells. J. Biol. Chem. 1976;251:3977–3984. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

