MicroRNA-687 Induced by Hypoxia-Inducible Factor-1 Targets Phosphatase and Tensin Homolog in Renal Ischemia-Reperfusion Injury
- PMID: 25587068
- PMCID: PMC4483585
- DOI: 10.1681/ASN.2014050463
MicroRNA-687 Induced by Hypoxia-Inducible Factor-1 Targets Phosphatase and Tensin Homolog in Renal Ischemia-Reperfusion Injury
Abstract
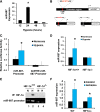
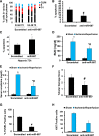
Ischemia-reperfusion injury contributes to tissue damage and organ failure in clinical settings, but the underlying mechanism remains elusive and effective therapies are still lacking. Here, we identified microRNA 687 (miR-687) as a key regulator and therapeutic target in renal ischemia-reperfusion injury. We show that miR-687 is markedly upregulated in the kidney during renal ischemia-reperfusion in mice and in cultured kidney cells during hypoxia. MiR-687 induction under these conditions was mediated by hypoxia-inducible factor-1 (HIF-1). Upon induction in vitro, miR-687 repressed the expression of phosphatase and tensin homolog (PTEN) and facilitated cell cycle progression and apoptosis. Blockade of miR-687 preserved PTEN expression and attenuated cell cycle activation and renal apoptosis, resulting in protection against kidney injury in mice. Collectively, these results unveil a novel HIF-1/miR-687/PTEN signaling pathway in ischemia-reperfusion injury that may be targeted for therapy.
Keywords: acute renal failure; cell death; hypoxia; ischemia-reperfusion; renal injury.
Copyright © 2015 by the American Society of Nephrology.
Figures


Comment in
-
Epigenetic Changes Induced by Hypoxia-Inducible Factor: a Long Way Still To Go as a Target for Therapy?J Am Soc Nephrol. 2015 Jul;26(7):1478-80. doi: 10.1681/ASN.2014121161. Epub 2015 Jan 13. J Am Soc Nephrol. 2015. PMID: 25587069 Free PMC article. No abstract available.
References
-
- Ambros V: The functions of animal microRNAs. Nature 431: 350–355, 2004 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials

